Related Questions
The number of unpaired electrons in the ground state of chromium is
1
6
7
2
Which of the following atoms has a non-spherical outermost orbital.
H
Li
Be
B
For a given energy level the number of orbitals is equal to
Davisson and Germer gave an experimental evidence for
wave nature of electron
particle nature of electron
particle nature of light
wave nature of light
Isotopes have different
arrangement of electrons
no. of P and e'
no. of neutrons
no. of electrons
The maximum number of electrons that can be accommodated by an atom in g-sub-energy level are
20
25
18
12
The fundamental particle that has least mass is
meson
alpha-particle
electron
neutron
The atomic structure of an atom of an element is Is . The element is
Cl-35
S-32
N-18
O-16
Among the following ions, which are has the highest paramagnetism?
Electronic configuration of is
Argon is isoelectronic with
The wavelength of an electron moving with a velocity
Which elements is represented by the following electronic configuration?
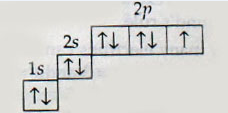
Nitrogen
Oxygen
Fluorine
Neon
Which of the following weighs the least?
1 g of
2 litres of at N.T.P.
molecules of
atoms of carbon
The correct set of quantum numbers for the impaired electron of fluorine atom is
| n | l | m |
| 2 | 1 | 0 |
| n | l | m |
| 2 | 1 | 1 |
| n | l | m |
| 3 | 1 | 1 |
| n | l | m |
| 3 | 0 | 0 |
Mass of positron is the same to that of
proton
meson
electron
neutron
The exchange of particles considered responsible for keeping the nucleons together are
meson
electron
positron
neutron
The atomic number of an element having the valence shell electronic configuration is
35
26
23
34
The orbitals with maximum number of possible orientation is:
s
d
f
p
If value of h is taken as , the de-Broglie wavelength of a particle of mass having velocity is
0.01 m
2 nm
0.1 nm
The angular momentum of the moving electron is equal to
The number of neutrons in the deuterium atom is
16
3
1
0
Cathode rays contain a stream of
Isotones have same
mass number
nuclear mass
no. of neutrons
no. of electrons
Frequency of the wave having wave number is
In Lyman series an electron jumps from higher energy level to
K energy level
M energy level
N energy level
L energy level
In the nucleus of there are
40 protons and 20 electrons
20 protons and 40 electrons
20 protons and 40 neutrons
20 protons and 20 neutrons
The size of nucleus is
rays are a stream of
He
electrons
positron
neutrons
The maximum number of electrons in a sub-energy level is equal to
