A coil is cut into two halves, the stiffness of cut coil will be
Double
Half
Same
None of these
Correct Answer :
A. Double
Related Questions
A beam is loaded as cantilever. If the load at the end is increased, the failure will occur
In the middle
At the tip below the load
At the support
Anywhere
Euler's formula holds good only for
Short columns
Long columns
Both short and long columns
Weak columns
Which of the following is correct?
Gauge pressure = Absolute pressure + Atmospheric pressure
Absolute pressure = Gauge pressure + Atmospheric pressure
Absolute pressure = Gauge pressure - Atmospheric pressure
Atmospheric pressure = Absolute pressure + Gauge pressure
Which of the following gas is mostly used in town for street and domestic lighting and heating?
Producer gas
Coal gas
Mond gas
Coke oven gas
The value of 1 mm of Hg is equal to
1.333 N/m2
13.33 N/m2
133.3 N/m2
1333 N/m2
The ultimate analysis of coal consists of the determination of the percentage of
Carbon
Hydrogen and nitrogen
Sulphur and ash
All of these
The deformation of a bar under its own weight compared to the deformation of same body subjected to a direct load equal to weight of the body is
Same
Double
Half
Four times
When it is indicated that a member is elastic, it means that when force is applied, it will
Not deform
Be safest
Stretch
Not stretch
Which of the following is the correct statement?
For a given compression ratio, both Otto and Diesel cycles have the same efficiency.
For a given compression ratio, Otto cycle is more efficient than Diesel cycle.
For a given compression ratio, Diesel cycle is more efficient than Otto cycle.
The efficiency of Otto or Diesel cycle has nothing to do with compression ratio.
Workdone during adiabatic expansion is given by (where p1 v1, T1 = Pressure, volume and temperature for the initial condition of gas, p2, v2, T2 = Corresponding values for the final condition of gas, R = Gas constant, and γ = Ratio of specific heats)
(p1 v1 - p2 v2)/(γ - 1)
[m R (T1 - T2)] /(γ - 1)
[m R T1/(γ - 1)][1 - (p2 v2 /p1 v1)]
All of these
The heat flows from a cold body to a hot body with the aid of an external source. This statement is given by
Kelvin
Joule
Clausis
Gay-Lussac
Within elastic limit, stress is
Inversely proportional to strain
Directly proportional to strain
Square root of strain
Equal to strain
A cylindrical section having no joint is known as
Joint less section
Homogeneous section
Perfect section
Seamless section
The efficiency of Stirling cycle is __________ Carnot cycle.
Greater than
Less than
Equal to
None of these
Diesel cycle consists of __________ processes.
Two constant volume and two isentropic
Two constant pressure and two isentropic
Two constant volume and two isothermal
One constant pressure, one constant volume and two isentropic
When the gas is cooled at constant pressure,
Its temperature increases but volume decreases
Its volume increases but temperature decreases
Both temperature and volume increases
Both temperature and volume decreases
Modulus of rigidity may be defined as the ratio of
Linear stress to lateral strain
Lateral strain to linear strain
Linear stress to linear strain
Shear stress to shear strain
The entropy __________ in an irreversible cyclic process.
Remains constant
Decreases
Increases
None of these
The heat energy stored in the gas and used for raising the temperature of the gas is known as
External energy
Internal energy
Kinetic energy
Molecular energy
If in the equation pvn = C, the value of n = ∝, then the process is called
Constant volume process
Adiabatic process
Constant pressure process
Isothermal process
The main cause for the irreversibility is
Mechanical and fluid friction
Unrestricted expansion
Heat transfer with a finite temperature difference
All of the above
Elasticity of Mild Steel specimen is defined by
Hookes law
Yield point
Plastic flow
Proof stress
A path 1-2-3 is given. A system absorbs 100 kJ as heat and does 60 kJ of work while along the path 1-4-3, it does 20 kJ of work. The heat absorbed during the cycle 1-4-3 is 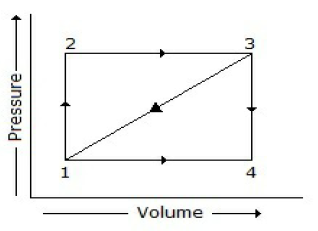
-140 kJ
-80 kJ
-40 kJ
+60 kJ
Carnot cycle consists of
Two constant volume and two isentropic processes
Two isothermal and two isentropic processes
Two constant pressure and two isentropic processes
One constant volume, one constant pressure and two isentropic processes
The fuel mostly used in cement industry and in metallurgical processes is
Wood charcoal
Bituminous coke
Pulverised coal
Coke
A composite shaft consisting of two stepped portions having spring constants K₁ and K₂ is held between two rigid supports at the ends. Its equivalent spring constant is
K₁ K₂
(K₁ + K₂)/ 2
(K₁ + K₂)/ K₁ K₂
K₁ K₂/ (K₁ + K₂)
In a tensile test, near the elastic limit zone, the
Tensile strain increases more quickly
Tensile strain decreases more quickly
Tensile strain increases in proportion to the stress
Tensile strain decreases in proportion to the stress
The kinetic energy per kg molecule of any gas at absolute temperature T is equal to (where Ru = Universal gas constant)
Ru × T
1.5 Ru × T
2 Ru × T
3 Ru × T
Which of the following cycles has maximum efficiency?
Rankine
Stirling
Carnot
Brayton
The polytropic index (n) is given by
log (p1p2)/log (v1v2)
log (p2/ p1)/log (v1/ v2)
log (v1/ v2)/ log (p1/p2)
log [(p1v1)/(p2v2)]
