A thin mild steel wire is loaded by adding loads in equal increments till it breaks. The extensions noted with increasing loads will behave as under
Uniform throughout
Increase uniformly
First increase and then decrease
Increase uniformly first and then increase rapidly
Correct Answer :
D. Increase uniformly first and then increase rapidly
Related Questions
The compression ratio for petrol engines is
3 to 6
5 to 8
10 to 20
15 to 30
For a beam, as shown in the below figure, the deflection at C is (where E = Young's modulus for the beam material, and I = Moment of inertia of the beam section.
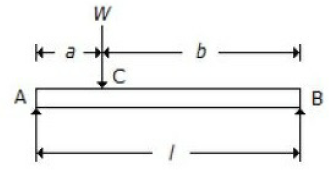
Wl3/48 EI
Wa²b²/3EIl
[Wa/(a√3) x EIl] x (l² - a²)3/2
5Wl3/384 EI
First law of thermodynamics deals with
Conservation of heat
Conservation of momentum
Conservation of mass
Conservation of energy
Shear modulus is the ratio of
Linear stress to linear strain
Linear stress to lateral strain
Volumetric strain to linear strain
Shear stress to shear strain
The state of stress at a point in a loaded member is shown in the below figure. The magnitude of maximum shear stress is
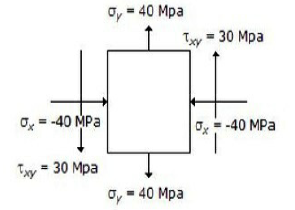
10 MPa
30 MPa
50 MPa
100 MPa
In a uniform bar, supported at one end in position, the maximum stress under self weight of bar shall occur at the
Middle of bar
Supported end
Bottom end
None of these
When a gas is heated, change takes place in
Pressure
Volume
Temperature
All of these
Which of the following statement is correct?
The increase in entropy is obtained from a given quantity of heat at a low temperature.
The change in entropy may be regarded as a measure of the rate of the availability or unavailability of heat for transformation into work.
The entropy represents the maximum amount of work obtainable per degree drop in temperature.
All of the above
The universal gas constant (or molar constant) of a gas is the product of
Molecular mass of the gas and the gas constant
Atomic mass of the gas and the gas constant
Molecular mass of the gas and the specific heat at constant pressure
Molecular mass of the gas and the specific heat at constant volume
The value of 1 mm of Hg is equal to
1.333 N/m2
13.33 N/m2
133.3 N/m2
1333 N/m2
The polytropic index (n) is given by
log (p1p2)/log (v1v2)
log (p2/ p1)/log (v1/ v2)
log (v1/ v2)/ log (p1/p2)
log [(p1v1)/(p2v2)]
Young's modulus is defined as the ratio of
Volumetric stress and volumetric strain
Lateral stress and lateral strain
Longitudinal stress and longitudinal strain
Shear stress to shear strain
If the radius of wire stretched by a load is doubled, then its Youngs modulus will be
Doubled
Halved
Becomes four times
None of the above
The efficiency of Diesel cycle approaches to Otto cycle efficiency when
Cut-off is increased
Cut-off is decreased
Cut-off is zero
Cut-off is constant
In a steady flow process, the ratio of
Heat transfer is constant
Work transfer is constant
Mass flow at inlet and outlet is same
All of these
Which of the following gas has the highest calorific value?
Coal gas
Producer gas
Mond gas
Blast furnace gas
When the expansion or compression takes place according to the law pvn = C, the process is known as
Isothermal process
Adiabatic process
Hyperbolic process
Polytropic process
High air-fuel ratio in gas turbines
Increases power output
Improves thermal efficiency
Reduces exhaust temperature
Do not damage turbine blades
According to Euler's column theory, the crippling load for a column of length (l) with one end fixed and the other end free is __________ the crippling load for a similar column hinged at both the ends.
Equal to
Less than
More than
None of these
A definite area or a space where some thermodynamic process takes place is known as
Thermodynamic system
Thermodynamic cycle
Thermodynamic process
Thermodynamic law
A column that fails due to direct stress, is called
Short column
Long column
Weak column
Medium column
When shear force at a point is zero, then bending moment is _________ at that point.
Zero
Minimum
Maximum
Infinity
Steam coal is a
Pulverised coal
Brown coal
Coking bituminous coal
Non-coking bituminous coal
In an ideal gas turbine plant, it is assumed that the compression and expansion processes are
Isothermal
Isentropic
Polytropic
None of these
The given figure shows the Mohr's circle of stress for two unequal and like principal stresses (σx and σy) acting at a body across two mutually perpendicular planes. The normal stress on an oblique section making an angle θ with the minor principle plane is given by
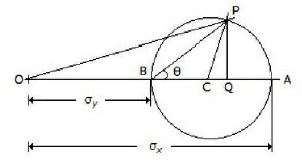
OC
OP
OQ
PQ
The pressure exerted by an ideal gas is ________ of the kinetic energy of all the molecules contained in a unit volume of gas.
One-half
One-third
Two-third
Three-fourth
The property of a material by virtue of which a body returns to its original, shape after removal of the load is called
Plasticity
Elasticity
Ductility
Malleability
The deformation of a bar under its own weight compared to the deformation of same body subjected to a direct load equal to weight of the body is
Same
Double
Half
Four times
One reversible heat engine operates between 1600 K and T2 K and another reversible heat engine operates between T2 K and 400 K. If both the engines have the same heat input and output, then temperature T2 is equal to
800 K
1000 K
1200 K
1400 K
When the gas is heated at constant volume, the heat supplied
Increases the internal energy of the gas and increases the temperature of the gas
Does some external work during expansion
Both (A) and (B)
None of these
