Consider the process A & B shown in the figure given below: In this case, it is possible that
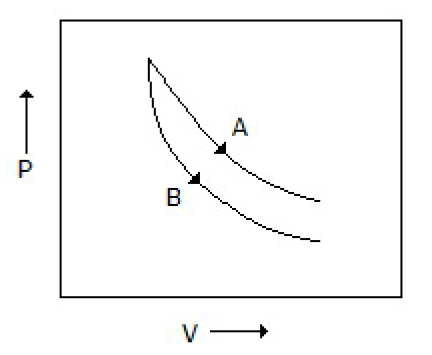
Both the processes are adiabatic
Both the processes are isothermal
Process A is isothermal while B is adiabatic
Process A is adiabatic while B is isothermal
Correct Answer :
C. Process A is isothermal while B is adiabatic
Related Questions
Change of heat content when one mole of compound is burnt in oxygen at constant pressure is called the
Calorific value
Heat of reaction
Heat of combustion
Heat of formation
At constant temperature and pressure, for one mole of a pure substance, the ratio of the free energy to the chemical potential is
Zero
One
Infinity
Negative
Solid and liquid phases of a substance are in equilibrium at the
Critical temperature
Melting point
Freezing point
Both (B) and (C)
At absolute zero temperature, the __________ of the gas is zero.
Pressure
Volume
Mass
None of these
Mollier diagram is a plot of
Temperature vs. enthalpy
Temperature vs. enthalpy
Entropy vs. enthalpy
Temperature vs. internal energy
In a homogeneous solution, the activity coefficient of a component depends upon the
Pressure
Composition
Temperature
All (A), (B) and (C)
In the equation, PVn = constant, if the value of n is in between 1 and y (i.e. Cp/Cv), then it represents a reversible __________ process.
Isometric
Polytropic
Isentropic
Isobaric
Which of the following is not an intensive property?
Volume
Density
Temperature
Pressure
When a gas in a vessel expands, its internal energy decreases. The process involved is
Reversible
Irreversible
Isothermal
Adiabatic
The enthalpy change when ammonia gas is dissolved in water is called the heat of
Solution
Formation
Dilution
Combustion
If the internal energy of an ideal gas decreases by the same amount as the work done by the system, then the
Process must be isobaric
Temperature must decrease
Process must be adiabatic
Both (B) and (C)
Steam undergoes isentropic expansion in a turbine from 5000 kPa and 400°C (entropy = 6.65 kJ/kg K) to 150 kPa) (entropy of saturated liquid = 1.4336 kJ/kg. K, entropy of saturated vapour = 7.2234 kJ/kg. K) The exit condition of steam is
Superheated vapour
Partially condensed vapour with quality of 0.9
Saturated vapour
Partially condensed vapour with quality of 0.1
Degree of freedom of a system consisting of a gaseous mixture of H2 and NH3 will be
0
1
2
3
Joule-Thomson co-efficient for a perfect gas is
Zero
Positive
Negative
None of these
As pressure approaches zero, the ratio of fugacity to pressure (f/P) for a gas approaches
Zero
Unity
Infinity
An indeterminate value
Which of the following is a widely used refrigerant in vapour compression refrigeration system (using large centrifugal compressor)?
Freon
Liquid sulphur dioxide
Methyl chloride
Ammonia
A solute distributes itself between two nonmiscible solvents in contact with each other in such a way that, at a constant temperature, the ratio of its concentrations in two layers is constant, irrespective of its total amount. This is
The distribution law
Followed from Margules equation
A corollary of Henry's law
None of these
All gases during throttling process at atmospheric temperature and pressure show a cooling effect except
CO2
H2
O2
N2
What happens in a reversible adiabatic compression?
Heating occurs
Cooling occurs
Pressure is constant
Temperature is constant
Ideal gas law is applicable at
Low T, low P
High T, high P
Low T, high P
High T, low P
For an ideal gas, the chemical potential is given by
RT d ln P
R d ln P
R d ln f
None of these
With increase in temperature, the atomic heat capacities of all solid elements
Increases
Decreases
Remains unchanged
Decreases linearly
The gas law (PV = RT) is true for an __________ change.
Isothermal
Adiabatic
Both (A) & (B)
Neither (A) nor (B)
Which of the following processes cannot be made reversible even under ideal condition of operation?
Free expansion of a gas
Compression of air in a compressor
Expansion of steam in a turbine
All (A), (B) & (C)
Which of the following is true for Virial equation of state?
Virial co-efficients are universal constants
Virial co-efficients 'B' represents three body interactions
Virial co-efficients are function of temperature only
For some gases, Virial equations and ideal gas equations are the same
Refrigeration cycle
Violates second law of thermodynamics
Involves transfer of heat from low temperature to high temperature
Both (A) and (B)
Neither (A) nor (B)
In an ideal solution, the activity of a component equals its
Mole fraction
Fugacity at the same temperature and pressure
Partial pressure
None of these
Cp - Cv = R is valid for __________ gases.
Ideal
Very high pressure
Very low temperature
All of the above
The expression for entropy change given by, ΔS = nR ln (V2/V1) + nCv ln (T2/T1) is valid for
Reversible isothermal volume change
Heating of a substance
Cooling of a substance
Simultaneous heating and expansion of an ideal gas
Air-refrigeration cycle
Is the most efficient of all refrigeration cycles
Has very low efficiency
Requires relatively large quantities of air to achieve a significant amount of refrigeration
Both (B) and (C)
