For the gaseous phase chemical reaction, C2H4(g) + H2O(g) ↔ C2H5OH(g), the equilibrium conversion does not depend on the
Steam to ethylene ratio
Temperature
Pressure
None of these
Correct Answer :
D. None of these
Related Questions
What happens in a reversible adiabatic expansion process?
Heating takes place
Cooling takes place
Pressure is constant
Temperature is constant
The partial pressure of each constituent present in an alloy is __________ the total vapor pressure exerted by the alloy.
Less than
Equal to
More than
Either (B) or (C); depends on the type of alloy
Gibbs-Duhem equation
States that n1dμ1 + n2dμ2 + ....njdμj = 0, for a system of definite composition at constant temperature and pressure
Applies only to binary systems
Finds no application in gas-liquid equilibria involved in distillation
None of these
If the vapour pressure at two temperatures of a solid phase in equilibrium with its liquid phase are known, then the latent heat of fusion can be calculated by the
Maxwell's equation
Clausius-Clapeyron Equation
Van Laar equation
Nernst Heat Theorem
Reduced pressure of a gas is the ratio of its
Pressure to critical pressure
Critical pressure to pressure
Pressure to pseudocritical pressure
Pseudocritical pressure to pressure
Enthalpy of a gas depends upon its
Temperature
Mass
Volume
Pressure
The enthalpy change when ammonia gas is dissolved in water is called the heat of
Solution
Formation
Dilution
Combustion
Entropy, which is a measure of the disorder of a system, is:
Independent of pressure
Independent of temperature
Zero at absolute zero temperature for a perfect crystalline substance
All (A), (B) & (C)
Entropy is a/an
State function
Macroscopic property
Extensive property
None of these
As the time is passing, entropy of the universe
Is increasing
Is decreasing
Remain constant
Data insufficient, can't be predicted
The variation of heat of reaction with temperature at constant pressure is given by the __________ law.
Kelvin's
Antoines
Kirchoffs
None of these
Consider the process A & B shown in the figure given below: In this case, it is possible that
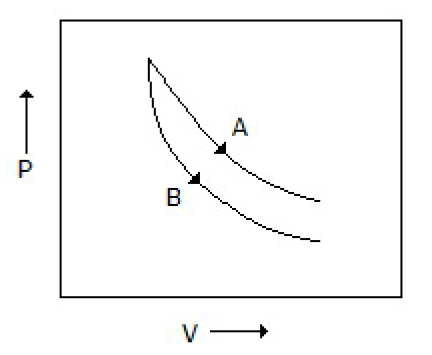
Both the processes are adiabatic
Both the processes are isothermal
Process A is isothermal while B is adiabatic
Process A is adiabatic while B is isothermal
In a homogeneous solution, the activity coefficient of a component depends upon the
Pressure
Composition
Temperature
All (A), (B) and (C)
Critical temperature is defined as the temperature above which a gas will
Not liquify (barring exceptions)
Immediately liquify
Never liquify however high the pressure may be
None of these
For an exothermic reaction
Only enthalpy change (ΔH) is negative
Only internal energy change (ΔE) is negative
Both ΔH and ΔE are negative
Enthalpy change is zero
For an ideal gas, the enthalpy
Increases with rise in pressure
Decreases with rise in pressure
Is independent of pressure
Is a path function
Which of the following is not a reversible process?
Expansion of an ideal gas against constant pressure
Atmospheric pressure vaporisation of water at 100°C
Solution of NaCl in water at 50°C
None of these
Degree of freedom of the system ice-watervapour will be
0
1
2
3
A refrigeration cycle is a reversed heat engine. Which of the following has the maximum value of the co-efficient of performance (COP) for a given refrigeration effect?
Vapor compression cycle using expansion valve
Air refrigeration cycle
Vapor compression cycle using expansion engine
Carnot refrigeration cycle
Clausius-Clapeyron equation is applicable to __________ equilibrium processes.
Solid-vapor
Solid-liquid
Liquid-vapor
All (A), (B) and (C)
Consider the reaction, C + O2 CO2; ΔH = - 94 kcal. What will be the value of ΔH for the reaction CO2 → C + O2?
-94 kcal
+94 kcal
> 94 kcal
< -94 kcal
Which of the following behaves most closely like an ideal gas?
He
N2
O2
H2
What is the ratio of adiabatic compressibility to isothermal compressibility?
1
< 1
> 1
>> 1
Pick out the undesirable property for a good refrigerant.
High thermal conductivity
Low freezing point
Large latent heat of vaporisation
High viscosity
Pick out the Clausius-Clapeyron equation from the following:
dP/dT = ΔH/TΔV
ln P = - (ΔH/RT) + constant
ΔF = ΔH + T [∂(ΔF)/∂T]P
None of these
For an isothermal reversible compression of an ideal gas
Only ΔE = 0
Only ΔH =0
ΔE = ΔH = 0
dQ = dE
The value of Joule-Thomson co-efficient, in case where cooling occurs after the throttling process is
0
∞
+ve
-ve
A closed system is cooled reversibly from 100°C to 50°C. If no work is done on the system
its internal energy (U) decreases and its entropy (S) increases
U and S both decreases
U decreases but S is constant
U is constant but S decreases
Co-efficient of Performance (COP) of a refrigerator is the ratio of the
Work required to refrigeration obtained
Refrigeration obtained to the work required
Lower to higher temperature
Higher to lower temperature
What happens in a reversible adiabatic compression?
Heating occurs
Cooling occurs
Pressure is constant
Temperature is constant
