Entropy, which is a measure of the disorder of a system, is:
Independent of pressure
Independent of temperature
Zero at absolute zero temperature for a perfect crystalline substance
All (A), (B) & (C)
Correct Answer :
C. Zero at absolute zero temperature for a perfect crystalline substance
Related Questions
Air-refrigeration cycle
Is the most efficient of all refrigeration cycles
Has very low efficiency
Requires relatively large quantities of air to achieve a significant amount of refrigeration
Both (B) and (C)
A solute distributes itself between two nonmiscible solvents in contact with each other in such a way that, at a constant temperature, the ratio of its concentrations in two layers is constant, irrespective of its total amount. This is
The distribution law
Followed from Margules equation
A corollary of Henry's law
None of these
Which is not a refrigerant?
SO2
NH3
CCl2F2
C2H4Cl2
The adiabatic throttling process of a perfect gas is one of constant enthalpy
In which there is a temperature drop
Which is exemplified by a non-steady flow expansion
Which can be performed in a pipe with a constriction
In which there is an increase in temperature
Which of the following units is not present in both the vapor compression refrigeration system and absorption refrigeration system?
Expansion valve
Condenser
Refrigerator
Compressor
In the equation, PVn = constant, if the value of n = ± ∞, then it represents a reversible __________ process.
Adiabatic
Isometric
Isentropic
Isothermal
For a multi-component system, the term chemical potential is equivalent to the
Molal concentration difference
Molar free energy
Partial molar free energy
Molar free energy change
Dry ice is
Moisture free ice
Solid helium
Solid carbon dioxide
None of these
Pick out the correct statement.
Like internal energy and enthalpy, the absolute value of standard entropy for elementary substances is zero
Melting of ice involves increase in enthalpy and a decrease in randomness
The internal energy of an ideal gas depends only on its pressure
Maximum work is done under reversible conditions
The internal energy of a gas obeying P (V - b) RT (where, b is a positive constant and has a constant Cv), depends upon its
Pressure
Volume
Temperature
All (A), (B) & (C)
It is desired to bring about a certain change in the state of a system by performing work on the system under adiabatic conditions.
The amount of work needed is path dependent
Work alone cannot bring out such a change of state
The amount of work needed is independent of path
More information is needed to conclude anything about the path dependence or otherwise of the work needed
Trouton's ratio is given by (where λb, = molal heat of vaporisation of a substance at its normal boiling point, kcal/kmol Tb = normal boiling point, °K)
λb/Tb
Tb/λb
√(λb/Tb)
√(Tb/λb)
Consider the process A & B shown in the figure given below: In this case, it is possible that
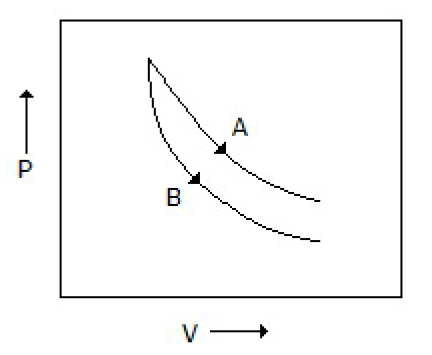
Both the processes are adiabatic
Both the processes are isothermal
Process A is isothermal while B is adiabatic
Process A is adiabatic while B is isothermal
Isobaric process means a constant process.
Temperature
Pressure
Volume
Entropy
Minimum number of phases that exists in a system is 1. Number of chemical species in a colloidal system is
1
2
3
4
Entropy change of mixing two liquid substances depends upon the
Molar concentration
Quantity (i.e. number of moles)
Both (A) and (B)
Neither (A) nor (B)
Isotherm on an enthalpy-concentration diagram, for an ideal solution will be a
Straight line
Sine curve
Parabola
Hyperbola
A cyclic engine exchanges heat with two reservoirs maintained at 100 and 300°C respectively. The maximum work (in J) that can be obtained from 1000 J of heat extracted from the hot reservoir is
349
651
667
1000
Change of heat content when one mole of compound is burnt in oxygen at constant pressure is called the
Calorific value
Heat of reaction
Heat of combustion
Heat of formation
Molar heat capacity of water in equilibrium with ice at constant pressure is __________ Kcal/kg mole. °K
0
∞
50
100
Pick out the wrong statement.
A closed system does not permit exchange of mass with its surroundings but may permit exchange of energy.
An open system permits exchange of both mass and energy with its surroundings
The term microstate is used to characterise an individual, whereas macro-state is used to designate a group of micro-states with common characteristics
None of the above
y = specific heat ratio of an ideal gas is equal to
Cp/Cv
Cp/(CP-R)
1 + (R/CV)
All (A), (B) and (C)
The first law of thermodynamics is a restatement of the law of conservation of
Mass
Energy
Momentum
None of these
Out of the following refrigeration cycles, which one has the minimum COP (Co-efficient of performance)?
Air cycle
Carnot cycle
Ordinary vapour compression cycle
Vapour compression with a reversible expansion engine
Chemical potential is a/an
Extensive property
Intensive property
Force which drives the chemical system to equilibrium
Both (B) and (C)
For a constant pressure reversible process, the enthalpy change (ΔH) of the system is
Cv.dT
Cp.dT
∫ Cp.dT
∫ Cv.dT
The enthalpy change when ammonia gas is dissolved in water is called the heat of
Solution
Formation
Dilution
Combustion
Heat of reaction at constant volume is identified with __________ change.
Enthalpy
Internal energy
Either (A) or (B)
Neither (A) nor (B)
Entropy change for an irreversible isolated system is
∞
0
< 0
> 0
As the temperature is lowered towards the absolute zero, the value of ∂(ΔF)/∂T, then approaches
Unity
Zero
That of the heat of reaction
Infinity
