Heat is added at constant temperature in an ideal __________ cycle.
Stirling
Brayton
Rankine
None of these
Correct Answer :
A. Stirling
Related Questions
There is a change in __________ during the phase transition.
Volume
Pressure
Temperature
All a, b & c
Pick out the extensive property out of the following.
Surface tension
Free energy
Specific heat
Refractive index
A change in state involving a decrease in entropy can be spontaneous, only if
It is exothermic
It is isenthalpic
It takes place isothermally
It takes place at constant volume
PVy = constant, holds good for an isentropic process, which is
Reversible and isothermal
Isothermal and irreversible
Reversible and adiabatic
Adiabatic and irreversible
If the molar heat capacities (Cp or Cv) of the reactants and products of a chemical reaction are identical, then, with the increase in temperature, the heat of reaction will
Increase
Decrease
Remain unaltered
Increase or decrease; depends on the particular reaction
To obtain integrated form of Clausius-Clapeyron equation, ln (P2/P1) = (ΔHV/R) (1/T1 - 1/T2) from the exact Clapeyron equation, it is assumed that the
Volume of the liquid phase is negligible compared to that of vapour phase
Vapour phase behaves as an ideal gas
Heat of vaporisation is independent of temperature
All (A), (B) & (C)
If the pressure on 100 c.c. of air is halved, then its volume (at the same temperature) would be __________ c.c.
100
50
205
200
COP of a refrigerator drawing 1 kW of power per ton of refrigeration is about
0.5
3.5
4.5
8.5
Gibbs-Duhem equation relates composition in liquid phase and the __________ at constant temperature & pressure.
Fugacity
Partial pressure
Activity co-efficient
All (A), (B), and (C)
Refrigeration capacity of a household refrigerator may be round about __________ tons.
0.15
1.5
4.5
6.5
For a real gas, the chemical potential is given by
RT d ln P
RT d ln f
R d ln f
None of these
In case of vapour compression refrigeration system, elevating the evaporator temperature (keeping the condenser temperature constant) results in
Enhanced COP
Decreased COP
No change in the value of COP
Increased or decreased COP; depending upon the type of refrigerant
Cv is given by
(∂E/∂T)V
(∂E/∂V)T
(∂E/∂P)V
(∂V/∂T)P
The value of Cp & Cv respectively for monatomic gases in Kcal/kg Mole.°K are
5 & 3
3.987 & 1.987
1.987 & 0.66
0.66 & 1.987
Clausius-Clapeyron Equation gives accurate result, when the
Vapour pressure is relatively low and the temperature does not vary over wide limits
Vapour obeys the ideal gas law and the latent heat of vaporisation is constant
Volume in the liquid state is negligible compared with that in the vapour state
All (A), (B) and (C)
A liquid under pressure greater than its vapour pressure for the temperature involved is called a __________ liquid.
Sub-cooled
Saturated
Non-solidifiable
None of these
A system undergoes a change from a given initial state to a given final state either by an irreversible process or by a reversible process, then (where, Δ S1 and Δ SR are the entropy changes of the system for the irreversible and reversible processes respectively)
Δ S1 is always < Δ SR
Δ S1 is sometimes > Δ SR
Δ S1 is always > Δ SR
Δ S1 is always = Δ SR
Pick out the wrong statement.
Minimum number of degree of freedom of a system is zero
Degree of freedom of a system containing a gaseous mixture of helium, carbon dioxide and hydrogen is 4
For a two phase system in equilibrium made up of four non-reacting chemical species, the number of degrees of freedom is 4
Enthalpy and internal energy change is zero during phase change processes like melting, vaporisation and sublimation
When pressure is applied on the system, ice ↔ water, then
Equilibrium cannot be established
More ice will be formed
More water will be formed
Evaporation of water will take place
Pick out the correct statement.
A real gas on expansion in vacuum gets heated up
An ideal gas on expansion in vacuum gets cooled
An ideal gas on expansion in vacuum gets heated up
A real gas on expansion in vacuum cools down whereas ideal gas remains unaffected
Standard temperature and pressure (S.T.P.) is
0°C and 750 mm Hg
15°C and 750 mm Hg
0°C and 1 kgf/cm2
15°C and 1 kgf/cm2
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
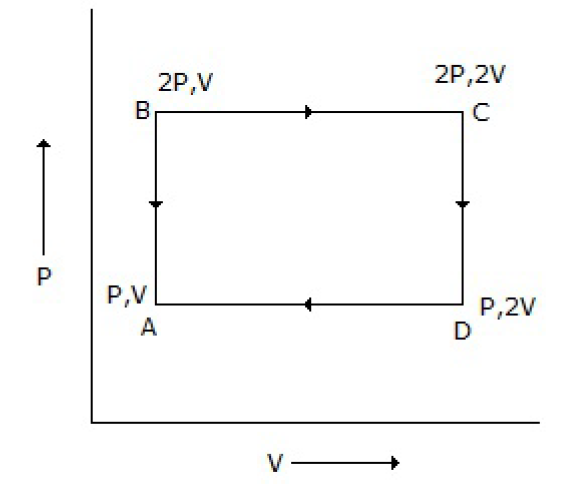
PV
2PV
PV/2
0
__________ decreases during adiabatic throttling of a perfect gas.
Entropy
Temperature
Enthalpy
Pressure
The value of Joule-Thomson co-efficient, in case where cooling occurs after the throttling process is
0
∞
+ve
-ve
Which of the following units is not present in both the vapor compression refrigeration system and absorption refrigeration system?
Expansion valve
Condenser
Refrigerator
Compressor
In the reaction, H2 +I2 2HI, addition of an inert gas will
Increase the partial pressure of H2
Increase the partial pressure of I2
Increase the total pressure and hence shift the equilibrium towards the right
Not affect the equilibrium conditions
The molar excess Gibbs free energy, gE, for a binary liquid mixture at T and P is given by, (gE/RT) = A . x1. x2, where A is a constant. The corresponding equation for ln y1, where y1 is the activity co-efficient of component 1, is
A . x22
Ax1
Ax2
Ax12
Refrigerants commonly used for domestic refrigerators are
Ethyl chloride or methyl chloride
Freon-12
Propane
NH3 or CO2
Gibbs-Helmholtz equation is
ΔF = ΔH + T [∂(ΔF)/∂T]P
ΔF = ΔH - TΔT
d(E - TS) T, V < 0
dP/dT = ΔHvap/T.ΔVvap
Dry ice is
Moisture free ice
Solid helium
Solid carbon dioxide
None of these
