An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
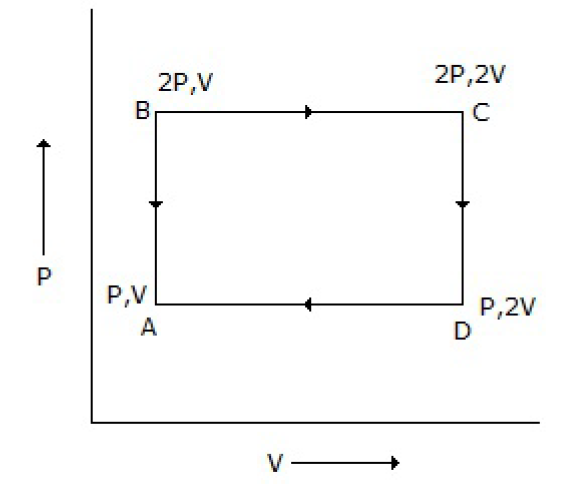
PV
2PV
PV/2
0
Correct Answer :
A. PV
Related Questions
Fugacity is a measure of the
Escaping tendencies of the same substance in different phases of a system
Relative volatility of a mixture of two miscible liquids
Behaviour of ideal gases
None of these
When pressure is applied on the system, ice ↔ water, then
Equilibrium cannot be established
More ice will be formed
More water will be formed
Evaporation of water will take place
In Joule-Thomson porous plug experiment, the
Enthalpy does not remain constant
Entire apparatus is exposed to surroundings
Temperature remains constant
None of these
Which of the following is an extensive property of a system?
Heat capacity
Molal heat capacity
Pressure
Concentration
dW and dq are not the exact differential, because q and W are
State functions
Path functions
Intensive properties
Extensive properties
Gibbs free energy at constant pressure and temperature under equilibrium conditions is
∞
0
Maximum
Minimum
Isentropic process means a constant __________ process.
Enthalpy
Pressure
Entropy
None of these
For an ideal gas, the activity co-efficient is
Directly proportional to pressure
Inversely proportional to pressure
Unity at all pressures
None of these
The rate at which a substance reacts is proportional to its active mass and the rate of a chemical reaction is proportional to the product of active masses of the reacting substances. This is the
Lewis-Randall rule
Statement of Van't Hoff Equation
Le-Chatelier's principle
None of these
Measurement of thermodynamic property of temperature is facilitated by __________ law of thermodynamics.
1st
Zeroth
3rd
None of these
Free energy changes for two reaction mechanism 'X' and 'Y are respectively - 15 and - 5 units. It implies that X is
Slower than Y
Faster than Y
Three times slower than Y
Three times faster than Y
If two gases have same reduced temperature and reduced pressure, then they will have the same
Volume
Mass
Critical temperature
None of these
Sublimation temperature of dry ice (solid CO2) is __________ °C.
-273
0
-78
5
Chemical potential of ith component of a system is given by
μi = (∂F/∂ni)T, P, ni
μi = (∂A/∂ni)T, P, ni
μi = (∂F/∂ni)T, P
μi = (∂A/∂ni)T, P
Pick out the Clausius-Clapeyron equation from the following:
dP/dT = ΔH/TΔV
ln P = - (ΔH/RT) + constant
ΔF = ΔH + T [∂(ΔF)/∂T]P
None of these
Melting of ice is an example of an __________ process.
Adiabatic
Isothermal
Isometric
None of these
The equation Tds = dE - PdV applies to
Single phase fluid of varying composition
Single phase fluid of constant composition
Open as well as closed systems
Both (B) and (C)
For the gaseous phase chemical reaction, C2H4(g) + H2O(g) ↔ C2H5OH(g), the equilibrium conversion does not depend on the
Steam to ethylene ratio
Temperature
Pressure
None of these
Van Laar equation deals with the activity coefficients in
Binary solutions
Ternary solutions
Azeotropic mixture only
None of these
Which of the following is a widely used refrigerant in vapour compression refrigeration system (using large centrifugal compressor)?
Freon
Liquid sulphur dioxide
Methyl chloride
Ammonia
The value of Cp & Cv respectively for monatomic gases in Kcal/kg Mole.°K are
5 & 3
3.987 & 1.987
1.987 & 0.66
0.66 & 1.987
Which of the following is Clausius-Clapeyron Equation for vaporisation of an ideal gas under the condition that the molar volume of liquid is negligible compared to that of the vapor?
d ln p/dt = Hvap/RT2
d ln p/dt = RT2/Hvap
dp/dt = RT2/Hvap
dp/dt = Hvap/RT2
Heat is added at constant pressure in an ideal __________ cycle.
Stirling
Brayton
Rankine
Both (B) and (C)
Free energy change of mixing two liquid substances is a function of the
Concentration of the constituents only
Quantities of the constituents only
Temperature only
All (A), (B) and (C)
Pick out the extensive property out of the following.
Surface tension
Free energy
Specific heat
Refractive index
What is the ratio of adiabatic compressibility to isothermal compressibility?
1
< 1
> 1
>> 1
If we increase the pressure on a substance (which is at its triple point), then the triple point
Increases
Decreases
Remains unchanged
May increase or decrease; depends on the substance
For an isothermal process, the internal energy of a gas
Increases
Decreases
Remains unchanged
Data insufficient, can't be predicted
High __________ is an undesirable property for a good refrigerant.
Specific heat
Latent heat of vaporisation
Viscosity
Specific vapor volume
With increase in reduced temperature, the fugacity co-efficient of a gas at constant reduced pressure
Increases
Decreases
Remain same
Decreases linearly
