For a real gas, the chemical potential is given by
RT d ln P
RT d ln f
R d ln f
None of these
Correct Answer :
B. RT d ln f
Related Questions
Second law of thermodynamics is concerned with the
Amount of energy transferred
Direction of energy transfer
Irreversible processes only
Non-cyclic processes only
Solid and liquid phases of a substance are in equilibrium at the
Critical temperature
Melting point
Freezing point
Both (B) and (C)
Compressibility factor-reduced pressure plot on reduced co-ordinates facilitates
Use of only one graph for all gases
Covering of wide range
Easier plotting
More accurate plotting
Entropy change for an irreversible isolated system is
∞
0
< 0
> 0
All gases except __________ shows a cooling effect during throttling process at atmospheric temperature and pressure.
Oxygen
Nitrogen
Air
Hydrogen
Tea kept in a thermos flask is vigorously shaken. If the tea is considered as a system, then its temperature will
Increase
Decrease
Remain unchanged
First fall and then rise
Critical compressibility factor for all substances
Are more or less constant (vary from 0.2 to 0.3)
Vary as square of the absolute temperature
Vary as square of the absolute pressure
None of these
Number of components (C), phase (P) and degrees of freedom (F) are related by Gibbs phase rule as
P + F - C = 2
C = P - F + 2
F = C - P - 2
P = F - C - 2
Gibbs-Duhem equation relates composition in liquid phase and the __________ at constant temperature & pressure.
Fugacity
Partial pressure
Activity co-efficient
All (A), (B), and (C)
Which is an example of closed system?
Air compressor
Liquid cooling system of an automobile
Boiler
None of these
Which of the following is Virial equation of state?
(p + a/V2)(V - b) = nRT
PV = nRT
PV = A + B/V + C/V2 + D/V3 + ...
None of these
Refrigerants commonly used for domestic refrigerators are
Ethyl chloride or methyl chloride
Freon-12
Propane
NH3 or CO2
Fundamental principle of refrigeration is based on the __________ law of thermodynamics.
Zeroth
First
Second
Third
Boiling of liquid is accompanied with increase in the
Vapor pressure
Specific Gibbs free energy
Specific entropy
All (A), (B) and (C)
The equation relating E, P, V and T which is true for all substances under all conditions is given by (∂E/∂V)T = T(∂P/∂T)H - P. This equation is called the
Maxwell's equation
Thermodynamic equation of state
Equation of state
Redlich-Kwong equation of state
During the phase transition, __________ changes.
Pressure
Volume
Temperature
All (A), (B) and (C)
In an isothermal process on an ideal gas, the pressure increases by 0.5 percent. The volume decreases by about __________ percent.
0.25
0.5
0.75
1
The internal energy of an incompressible fluid depends upon its
Pressure
Temperature
Both (A) & (B)
Neither (A) nor (B)
In Joule-Thomson porous plug experiment, the
Enthalpy does not remain constant
Entire apparatus is exposed to surroundings
Temperature remains constant
None of these
The effect of changing the evaporator temperature on COP as compared to that of changing the condenser temperature (in vapour compression refrigeration system) is
Less pronounced
More pronounced
Equal
Data insufficient, can't be predicted
Which of the following is not a reversible process?
Expansion of an ideal gas against constant pressure
Atmospheric pressure vaporisation of water at 100°C
Solution of NaCl in water at 50°C
None of these
Dry ice is
Moisture free ice
Solid helium
Solid carbon dioxide
None of these
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
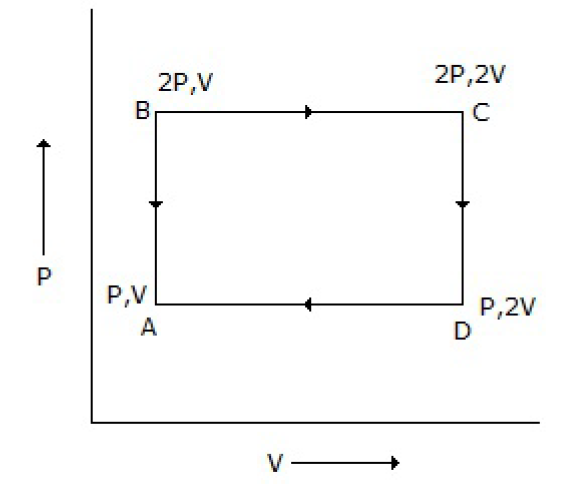
PV
2PV
PV/2
0
When dilute aqueous solutions of two salts are mixed, the process is associated with
Decrease in temperature
Increase in temperature
No change in temperature
Change in temperature which is a function of composition
In the ammonia synthesis reaction, N2 + 3H2 2NH3 + 22.4 kcal, the formation of NH3 will be favoured by
High temperature
Low pressure
Low temperature only
Both low temperature and high pressure
Adiabatic compression of a saturated water vapour makes it
Supersaturated
Superheated
Both (A) and (B)
Neither (A) nor (B)
In the equation, PVn = constant, if the value of n = ± ∞, then it represents a reversible __________ process.
Adiabatic
Isometric
Isentropic
Isothermal
The second law of thermodynamics states that
The energy change of a system undergoing any reversible process is zero
It is not possible to transfer heat from a lower temperature to a higher temperature
The total energy of system and surrounding remains the same
None of the above
An ideal liquid refrigerant should
Not have a sub-atmospheric vapour pressure at the temperature in the refrigerator coils
Not have unduly high vapour pressure at the condenser temperature
Both (A) and (B)
Have low specific heat
The following heat engine produces power of 100,000 kW. The heat engine operates between 800 K and 300 K. It has a thermal efficiency equal to 50% of that of the Carnot engine for the same temperature. The rate at which heat is absorbed from the hot reservoir is
100,000 kW
160,000 kW
200,000 kW
320,000 kW
