The atomic mass of nitrogen is __________ oxygen.
Equal to
Less than
More than
None of these
Correct Answer :
B. Less than
Related Questions
In the below figure, curve D represents_________.
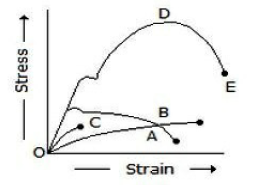
Mild steel
Cast iron
Concrete
Bone of these
Carbonisation of coal consists of
Drying and crushing the coal to a fine powder
Moulding the finely ground coal under pressure with or without a binding material
Heating the wood with a limited supply of air to temperature not less than 280°C
None of the above
The bending stress in a beam is _________ section modulus.
Inversely proportional to two times
Directly proportional to
Inversely proportional to
None of these
When a body is subjected to two equal and opposite pushes, as a result of which the body tends to reduce its length, then
The stress and strain induced is compressive
The stress and strain induced is tensile
Both A and B is correct
None of these
For which material the Poisson's ratio is more than unity
Steel
Copper
Aluminium
None of the above
Tensile strength of a material is obtained by dividing the maximum load during the test by the
Area at the time of fracture
Original cross-sectional area
Average of (A) and (B)
Minimum area after fracture
One kg of hydrogen requires 8 kg of oxygen and produces
1 kg of water
7 kg of water
8 kg of water
9 kg of water
If a material expands freely due to heating it will develop
Thermal stresses
Tensile stress
Bending
No stress
During which of the following process does heat rejection takes place in Carnot cycle?
Isothermal expansion
Isentropic expansion
Isothermal compression
Isentropic compression
The fuel mostly used in steam boilers is
Brown coal
Peat
Coking bituminous coal
Non-coking bituminous coal
The neutral axis of the cross-section a beam is that axis at which the bending stress is
Zero
Minimum
Maximum
Infinity
Reversed Joule cycle is known as
Carnot cycle
Bell-Coleman cycle
Rankine cycle
Stirling cycle
The ideal efficiency of a Brayton cycle without regeneration, with increase in pressure ratio will
Increase
Decrease
Remain unchanged
Increase/decrease depending on application
Strain is equal to (where l = Original length, and δl = Change in length)
l/δl
δl/l
l.δl
l + δl
The property of a material by virtue of which a body returns to its original, shape after removal of the load is called
Plasticity
Elasticity
Ductility
Malleability
Principal planes are planes having
Maximum shear stress
No shear stress
Minimum shear stress
None of the above
A body is subjected to a tensile stress of 1200 MPa on one plane and another tensile stress of 600 MPa on a plane at right angles to the former. It is also subjected to a shear stress of 400 MPa on the same planes. The maximum normal stress will be
400 MPa
500 MPa
900 MPa
1400 MPa
The behaviour of a perfect gas, undergoing any change in the variables which control physical properties, is governed by
Boyle's law
Charles' law
Gay-Lussac law
All of these
The stress developed in a material at breaking point in extension is called
Breaking stress
Fracture stress
Yield point stress
Ultimate tensile stress
Select the wrong statement
A Joule cycle consists of two constant volume and two isentropic processes.
An Otto cycle consists of two constant volume and two isentropic processes.
An Ericsson cycle consists of two constant pressure and two isothermal processes.
All of the above
The gas constant (R) is equal to the
Sum of two specific heats
Difference of two specific heats
Product of two specific heats
Ratio of two specific heats
The stress at which extension of the material takes place more quickly as compared to the increase in load is called
Elastic point of the material
Plastic point of the material
Breaking point of the material
Yielding point of the material
Producer gas is obtained by
Partial combustion of coal, coke, anthracite coal or charcoal in a mixed air steam blast
Carbonisation of bituminous coal
Passing steam over incandescent coke
Passing air and a large amount of steam over waste coal at about 650°C
The air standard efficiency of an Otto cycle is given by (where r = Compression ratio, and γ = Ratio of specific heats)
1 - rγ - 1
1 + rγ - 1
1 - (1/ rγ - 1)
1 + (1/ rγ - 1)
Otto cycle consists of
Two constant volume and two isentropic processes
Two constant pressure and two isentropic processes
Two constant volume and two isothermal processes
One constant pressure, one constant volume and two isentropic processes
The stress induced in a body due to suddenly applied load compared to when it is applied gradually is
Same
Half
Two times
Four times
The distance between the centres of the rivets in adjacent rows of zigzag riveted joint is known as
Pitch
Back pitch
Diagonal pitch
Diametric pitch
The materials having same elastic properties in all directions are called
Ideal materials
Uniform materials
Isotropic materials
Piratical materials
Which of the following statement is correct?
The increase in entropy is obtained from a given quantity of heat at a low temperature.
The change in entropy may be regarded as a measure of the rate of the availability or unavailability of heat for transformation into work.
The entropy represents the maximum amount of work obtainable per degree drop in temperature.
All of the above
The relation between equivalent length (L) and actual length (l) of a column for both ends fixed is
L = l/2
L = l/√2
L = l
L = 2l
