The rivets are used for __________ fastenings.
Permanent
Temporary
Semi-permanent
None of these
Correct Answer :
A. Permanent
Related Questions
The smallest quantity of a substance, which can exist by itself in a chemically recognizable form is known as
Element
Compound
Atom
Molecule
The value of specific heat at constant pressure (cp) is __________ that of at constant volume (cv).
Less than
Equal to
More than
None of these
A circular shaft fixed at, A has diameter D for half of its length and diameter D/2 over the other half, as shown in the below figure. If the rotation of B relative to A is 0.1 radian, the rotation of C relative to B will be
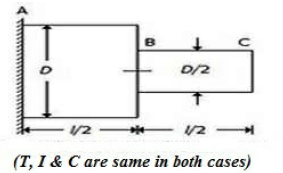
0.4 radian
0.8 radian
1.6 radian
3.2 radian
A hollow shaft of same cross-section area as compared to a solid shaft transmit
Same torque
Less torque
More torque
Unpredictable
Those substances which have so far not been resolved by any means into other substances of simpler form are called
Elements
Compounds
Atoms
Molecules
The natural solid fuel is
Wood
Coke
Anthracite coal
Pulverised coal
Proof resilience per material is known as
Resilience
Proof resilience
Modulus of resilience
Toughness
The absolute zero pressure will be
When molecular momentum of the system becomes zero
At sea level
At the temperature of - 273 K
At the centre of the earth
The energy stored in a body when strained within elastic limit is known as
Resilience
Proof resilience
Strain energy
Impact energy
Strain re-setters are used to
Measure shear strain
Measure linear strain
Measure volumetric strain
Relieve strain
A column that fails due to direct stress, is called
Short column
Long column
Weak column
Medium column
The work ratio of simple gas turbine cycle depends upon
Maximum cycle temperature
Minimum cycle temperature
Pressure ratio
All of these
Which of the following statement is correct?
The increase in entropy is obtained from a given quantity of heat at a low temperature.
The change in entropy may be regarded as a measure of the rate of the availability or unavailability of heat for transformation into work.
The entropy represents the maximum amount of work obtainable per degree drop in temperature.
All of the above
Carnot cycle has maximum efficiency for
Petrol engine
Diesel engine
Reversible engine
Irreversible engine
When a gas is heated at constant volume
Its temperature will increase
Its pressure will increase
Both temperature and pressure will increase
Neither temperature nor pressure will increase
In a free expansion process,
W1 - 2 = 0
Q1 - 2 = 0
dU = 0
All of these
If a material expands freely due to heating it will develop
Thermal stresses
Tensile stress
Bending
No stress
The change in the unit volume of a material under tension with increase in its Poisson's ratio will
Increase
Decrease
Remain same
Increase initially and then decrease
The distillation carried out in such a way that the liquid with the lowest boiling point is first evaporated and recondensed, then the liquid with the next higher boiling point is then evaporated and recondensed, and so on until all the available liquid fuels are separately recovered in the sequence of their boiling points. Such a process is called
Cracking
Carbonisation
Fractional distillation
Full distillation
The tensile strength of the welded joint for double fillet is (where s = Leg or size of the weld, l = Length of weld, and σt = Allowable tensile stress for weld metal)
0.5 s.l.σt
s.l.σt
√2 s.l.σt
2.s.l.σt
The gas in cooling chamber of a closed cycle gas turbine is cooled at
Constant volume
Constant temperature
Constant pressure
None of these
For the same compression ratio, the efficiency of dual combustion cycle is
Greater than Diesel cycle and less than Otto cycle
Less than Diesel cycle and greater than Otto cycle
Greater than Diesel cycle
Less than Diesel cycle
The moment of resistance of a balanced reinforced concrete beam is based on the stresses in
Steel only
Concrete only
Steel and concrete both
None of these
The value of gas constant (R) in S. I. units is
0.287 J/kgK
2.87 J/kgK
28.7 J/kgK
287 J/kgK
A cylindrical section having no joint is known as
Joint less section
Homogeneous section
Perfect section
Seamless section
The following cycle is used for air craft refrigeration
Brayton cycle
Joule cycle
Carnot cycle
Reversed Brayton cycle
The heat energy stored in the gas and used for raising the temperature of the gas is known as
External energy
Internal energy
Kinetic energy
Molecular energy
Which of the following is an irreversible cycle?
Carnot
Stirling
Ericsson
None of the above
Which of the following statement is correct?
The heat and work are boundary phenomena
The heat and work represent the energy crossing the boundary of the system
The heat and work are path functions
All of the above
Which of the following materials is most elastic?
Rubber
Plastic
Brass
Steel
