A key is subjected to side pressure as well at shearing forces. These pressures are called
Bearing stresses
Fatigue stresses
Crushing stresses
Resultant stresses
Correct Answer :
A. Bearing stresses
Related Questions
The volumetric or molar specific heat at constant pressure is the product of
Molecular mass of the gas and the specific heat at constant volume
Atomic mass of the gas and the gas constant
Molecular mass of the gas and the gas constant
None of the above
The efficiency and work ratio of a simple gas turbine cycle are
Low
Very low
High
Very high
The stress induced in a body due to suddenly applied load compared to when it is applied gradually is
Same
Half
Two times
Four times
The property of a material by virtue of which a body returns to its original, shape after removal of the load is called
Plasticity
Elasticity
Ductility
Malleability
The efficiency of the dual combustion cycle for the same compression ratio is __________ Diesel cycle.
Greater than
Less than
Equal to
None of these
If Kh is the torque resisting capacity of a hollow shaft and Ks is that of a solid shaft, of the same material, length and weight. Then,
Kh > Ks
Kh < Ks
Kh = Ks
None of these
The strain energy stored in a body due to suddenly applied load compared to when it is applied gradually is
Same
Twice
Four times
Eight times
When a gas is heated at constant volume
Its temperature will increase
Its pressure will increase
Both temperature and pressure will increase
Neither temperature nor pressure will increase
Flow stress corresponds to
Fluids in motion
Breaking point
Plastic deformation of solids
Rupture stress
Which of the following cycles is not a reversible cycle?
Carnot
Ericsson
Stirling
None of the above
An adiabatic process is one in which
No heat enters or leaves the gas
The temperature of the gas changes
The change in internal energy is equal to the mechanical workdone
All of the above
The value of Poisson's ratio for steel is between
0.01 to 0.1
0.23 to 0.27
0.25 to 0.33
0.4 to 0.6
The strain energy stored in a solid circular shaft in torsion, subjected to shear stress (τ), is: (Where, G = Modulus of rigidity for the shaft material)
τ²/ 2G × Volume of shaft
τ/ 2G × Volume of shaft
τ²/ 4G × Volume of shaft
τ/ 4G × Volume of shaft
A close cycle gas turbine gives __________ efficiency as compared to an open cycle gas turbine.
Same
Lower
Higher
None of these
The columns whose slenderness ratio is less than 80, are known as
Short columns
Long columns
Weak columns
Medium columns
The ideal efficiency of a Brayton cycle with regeneration, with increase in pressure ratio will
Increase
Decrease
Remain unchanged
Increase/decrease depending on application
The given figure shows the Mohr's circle of stress for two unequal and like principal stresses (σx and σy) acting at a body across two mutually perpendicular planes. The normal stress on an oblique section making an angle θ with the minor principle plane is given by
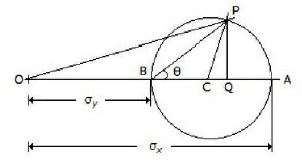
OC
OP
OQ
PQ
If both Stirling and Carnot cycles operate within the same temperature limits, then efficiency of Stirling cycle as compared to Carnot cycle
More
Less
Equal
Depends on other factors
A body is subjected to a tensile stress of 1200 MPa on one plane and another tensile stress of 600 MPa on a plane at right angles to the former. It is also subjected to a shear stress of 400 MPa on the same planes. The maximum normal stress will be
400 MPa
500 MPa
900 MPa
1400 MPa
Percentage reduction in area performing tensile test on cast iron may be of the order of
50 %
25 %
20 %
30 %
In an extensive property of a thermodynamic system
Extensive heat is transferred
Extensive work is done
Extensive energy is utilised
None of these
The ratio of maximum shear stress developed in a rectangular beam and a circular beam of the same cross-sectional area is
2/3
3/4
1
9/8
The state of stress at a point in a loaded member is shown in the below figure. The magnitude of maximum shear stress is
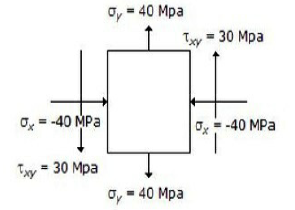
10 MPa
30 MPa
50 MPa
100 MPa
The total elongation produced in a bar of uniform section hanging vertically downwards due to its own weight is equal to that produced by a weight
Of same magnitude as that of bar and applied at the lower end
Half the weight of bar applied at lower end
Half of the square of weight of bar applied at lower end
One fourth of weight of bar applied at lower end
Which of the following is the correct statement?
All the reversible engines have the same efficiency.
All the reversible and irreversible engines have the same efficiency.
Irreversible engines have maximum efficiency.
All engines are designed as reversible in order to obtain maximum efficiency.
The fuel mostly used in steam boilers is
Brown coal
Peat
Coking bituminous coal
Non-coking bituminous coal
The weakest section of a diamond riveting is the section which passes through
The first row
The second row
The central row
One rivet hole of the end row
One kg of ethylene (C2H4) requires 2 kg of oxygen and produces 22/7 kg of carbon dioxide and __________ kg of water or steam.
9/7
11/7
7/4
1/4
In compression test, the fracture in cast iron specimen would occur along
The axis of load
An oblique plane
At right angles to the axis of specimen
Would not occur
The ratio of molar specific heats for mono-atomic gas is
1
1.4
1.67
1.87
