The value of Poisson's ratio for steel is between
0.01 to 0.1
0.23 to 0.27
0.25 to 0.33
0.4 to 0.6
Correct Answer :
C. 0.25 to 0.33
Related Questions
The heat and work are mutually convertible. This statement is called __________ law of thermodynamics.
Zeroth
First
Second
Third
Which of the following materials is most elastic?
Rubber
Plastic
Brass
Steel
One kg of hydrogen requires 8 kg of oxygen and produces
1 kg of water
7 kg of water
8 kg of water
9 kg of water
Strain energy is the
Energy stored in a body when strained within elastic limits
Energy stored in a body when strained up to the breaking of a specimen
Maximum strain energy which can be stored in a body
Proof resilience per unit volume of a material
For a perfect gas, according to Boyle's law (where p = Absolute pressure, v = Volume, and T = Absolute temperature)
p v = constant, if T is kept constant
v/T = constant, if p is kept constant
p/T = constant, if v is kept constant
T/p = constant, if v is kept constant
Reversed joule cycle is called
Carnot cycle
Rankine cycle
Brayton cycle
Bell Coleman cycle
A beam is loaded as cantilever. If the load at the end is increased, the failure will occur
In the middle
At the tip below the load
At the support
Anywhere
If the radius of wire stretched by a load is doubled, then its Youngs modulus will be
Doubled
Halved
Becomes four times
None of the above
The temperature at which the volume of a gas becomes zero is called
Absolute scale of temperature
Absolute zero temperature
Absolute temperature
None of these
According to Kelvin-Planck's statement of second law of thermodynamics,
It is impossible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
It is possible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
It is impossible to construct a device which operates in a cyclic process and produces no effect other than the transfer of heat from a cold body to a hot body
None of the above
The extremeties of any diameter on Mohr's circle represent
Principal stresses
Normal stresses on planes at 45°
Shear stresses on planes at 45°
Normal and shear stresses on a plane
The kinetic energy per kg molecule of any gas at absolute temperature T is equal to (where Ru = Universal gas constant)
Ru × T
1.5 Ru × T
2 Ru × T
3 Ru × T
Modulus of rigidity may be defined as the ratio of
Linear stress to lateral strain
Lateral strain to linear strain
Linear stress to linear strain
Shear stress to shear strain
The efficiency of a gas turbine is given by
(Net work output)/(Workdone by the turbine)
(Net work output)/(Heat supplied)
(Actual temperature drop)/(Isentropic temperature drop)
(Isentropic increase in temperature)/(Actual increase in temperature)
The sum of internal energy (U) and the product of pressure and volume (p.v) is known as
Workdone
Entropy
Enthalpy
None of these
The stress induced in a body, when suddenly loaded, is ________ the stress induced when the same load is applied gradually.
Equal to
One-half
Twice
Four times
The behaviour of a perfect gas, undergoing any change in the variables which control physical properties, is governed by
Boyle's law
Charles' law
Gay-Lussac law
All of these
In an extensive property of a thermodynamic system
Extensive heat is transferred
Extensive work is done
Extensive energy is utilised
None of these
The work ratio of a gas turbine plant is given by
(Net work output)/(Workdone by the turbine)
(Net work output)/(Heat supplied)
(Actual temperature drop)/(Isentropic temperature drop)
(Isentropic increase in temperature)/(Actual increase in temperature)
The unit of Young's modulus is
mm/mm
kg/cm
Kg
kg/cm²
In closed cycle gas turbine, the air is compressed
Isothermally
Isentropically
Polytropically
None of these
In a reversible adiabatic process, the ratio of T1/T2 is equal to
(p2/p1)γ - 1/ γ
(p1/p2)γ - 1/ γ
(v2/v1)γ - 1/ γ
(v1/v2)γ - 1/ γ
Which of the following is a proper sequence?
Proportional limit, elastic limit, yielding, failure
Elastic limit, proportional limit, yielding, failure
Yielding, proportional limit, elastic limit, failure
None of the above
The atomic mass of oxygen is
12
14
16
32
The heat absorbed or rejected by the working substance is given by (where ds = Increase or decrease of entropy, T = Absolute temperature, and dQ = Heat absorbed or rejected)
δQ = T.ds
δQ = T/ds
dQ = ds/T
None of these
If in the equation pvn = C, the value of n = ∝, then the process is called
Constant volume process
Adiabatic process
Constant pressure process
Isothermal process
A hollow shaft of same cross-section area as compared to a solid shaft transmit
Same torque
Less torque
More torque
Unpredictable
A path 1-2-3 is given. A system absorbs 100 kJ as heat and does 60 kJ of work while along the path 1-4-3, it does 20 kJ of work. The heat absorbed during the cycle 1-4-3 is 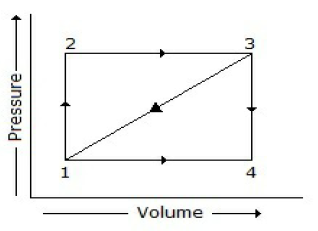
-140 kJ
-80 kJ
-40 kJ
+60 kJ
One kg of carbon monoxide requires 4/7 kg of oxygen and produces
11/3 kg of carbon dioxide gas
7/3 kg of carbon monoxide gas
11/7 kg of carbon dioxide gas
8/3 kg of carbon monoxide gas
The smallest quantity of a substance, which can exist by itself in a chemically recognizable form is known as
Element
Compound
Atom
Molecule
