The volumetric or molar specific heat at constant pressure is the product of
Molecular mass of the gas and the specific heat at constant volume
Atomic mass of the gas and the gas constant
Molecular mass of the gas and the gas constant
None of the above
Correct Answer :
D. None of the above
Related Questions
When a gas is heated at constant volume
Its temperature will increase
Its pressure will increase
Both temperature and pressure will increase
Neither temperature nor pressure will increase
A fletched beam is used to
Change the shape of the beam
Effect the saving in material
Equalise the strength in tension and compression
Increase the cross-section of the beam
The maximum stress produced in a bar of tapering section is at
Smaller end
Larger end
Middle
Anywhere
The pull required to tear off the plate per pitch length is (where p = Pitch of rivets, t = Thickness of plates, and σt, τ and σc = Permissible tensile, shearing and crushing stresses respectively)
(p - 2d) t × σc
(p - d) t × τ
(p - d) t × σt
(2p - d) t × σt
The efficiency of Diesel cycle with decrease in cut-off
Increases
Decreases
First increases and then decreases
First decreases and then increases
The columns whose slenderness ratio is less than 80, are known as
Short columns
Long columns
Weak columns
Medium columns
The heating of a gas at constant pressure is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
When a bar is subjected to a change of temperature and its deformation is prevented, the stress induced in the bar is
Tensile stress
Compressive stress
Shear stress
Thermal stress
When a body is subjected to a direct tensile stress (σx) in one plane accompanied by a simple shear stress (τxy), the maximum shear stress is
(σx/2) + (1/2) × √(σx² + 4 τ²xy)
(σx/2) - (1/2) × √(σx² + 4 τ²xy)
(σx/2) + (1/2) × √(σx² - 4 τ²xy)
(1/2) × √(σx² + 4 τ²xy)
A process of heating crude oil to a high temperature under a very high pressure to increase the yield of lighter distillates, is known as
Cracking
Carbonisation
Fractional distillation
Full distillation
For a beam, as shown in the below figure, when the load W is applied in the centre of the beam, the maximum deflection is
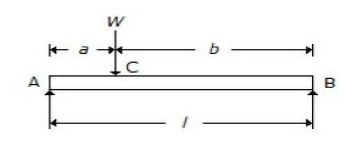
Wl3 / 48EI
5Wl3 / 384EI
Wl3 / 392EI
Wl3 / 384EI
The increase in entropy of a system represents
Increase in availability of energy
Increase in temperature
Decrease in pressure
Degradation of energy
The pull required to crush the rivet per pitch length is
p.t.σt
d.t.σc
π/4 × d² × σt
π/4 × d² × σc
Tensile strength of a material is obtained by dividing the maximum load during the test by the
Area at the time of fracture
Original cross-sectional area
Average of (A) and (B)
Minimum area after fracture
The main cause for the irreversibility is
Mechanical and fluid friction
Unrestricted expansion
Heat transfer with a finite temperature difference
All of the above
If percentage reduction in area of a certain specimen made of material 'A' under tensile test is 60% and the percentage reduction in area of a specimen with same dimensions made of material 'B' is 40%, then
The material A is more ductile than material B
The material B is more ductile than material A
The ductility of material A and B is equal
The material A is brittle and material B is ductile
Which of the following is correct?
Gauge pressure = Absolute pressure + Atmospheric pressure
Absolute pressure = Gauge pressure + Atmospheric pressure
Absolute pressure = Gauge pressure - Atmospheric pressure
Atmospheric pressure = Absolute pressure + Gauge pressure
A cantilever beam is one which is
Fixed at both ends
Fixed at one end and free at the other end
Supported at its ends
Supported on more than two supports
A key is subjected to side pressure as well at shearing forces. These pressures are called
Bearing stresses
Fatigue stresses
Crushing stresses
Resultant stresses
The heating of gas at constant volume is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
The __________ is obtained when carbonisation of coal is carried out at 500° to 700° C.
Soft coal
Hard coal
Pulverised coal
Bituminous coal
A cycle consisting of __________ and two isothermal processes is known as Stirling cycle.
Two constant pressure
Two constant volume
Two isentropic
One constant pressure, one constant volume
The strain energy stored in a solid circular shaft subjected to shear stress (τ), is: (Where G = Modulus of rigidity for the shaft material)
τ²/ 2G × Volume of shaft
τ/ 2G × Volume of shaft
τ²/ 4G × Volume of shaft
τ/ 4G × Volume of shaft
The volumetric or molar specific heat at constant pressure is the product of
Molecular mass of the gas and the specific heat at constant volume
Atomic mass of the gas and the gas constant
Molecular mass of the gas and the gas constant
None of the above
The natural petroleum may be separated into
Petrol
Kerosene
Fuel oil
Lubricating oil
The absolute zero pressure can be attained at a temperature of
0°C
273°C
273 K
None of these
Otto cycle consists of
Two constant volume and two isentropic processes
Two constant pressure and two isentropic processes
Two constant volume and two isothermal processes
One constant pressure, one constant volume and two isentropic processes
The air standard efficiency of an Otto cycle is given by (where r = Compression ratio, and γ = Ratio of specific heats)
1 - rγ - 1
1 + rγ - 1
1 - (1/ rγ - 1)
1 + (1/ rγ - 1)
When gas is heated at constant pressure, the heat supplied is utilised in
Increasing the internal energy of gas
Doing some external work
Increasing the internal energy of gas and also for doing some external work
None of the above
The entropy may be expressed as a function of
Pressure and temperature
Temperature and volume
Heat and work
All of these
