According to Kelvin-Planck's statement of second law of thermodynamics,
It is impossible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
It is possible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
It is impossible to construct a device which operates in a cyclic process and produces no effect other than the transfer of heat from a cold body to a hot body
None of the above
Correct Answer :
A. It is impossible to construct an engine working on a cyclic process, whose sole purpose is to convert heat energy into work
Related Questions
Workdone during adiabatic expansion is given by (where p1 v1, T1 = Pressure, volume and temperature for the initial condition of gas, p2, v2, T2 = Corresponding values for the final condition of gas, R = Gas constant, and γ = Ratio of specific heats)
(p1 v1 - p2 v2)/(γ - 1)
[m R (T1 - T2)] /(γ - 1)
[m R T1/(γ - 1)][1 - (p2 v2 /p1 v1)]
All of these
The neutral axis of the cross-section a beam is that axis at which the bending stress is
Zero
Minimum
Maximum
Infinity
When a system changes its state from one equilibrium state to another equilibrium state, then the path of successive states through which the system has passed, is known as
Thermodynamic law
Thermodynamic process
Thermodynamic cycle
None of these
Which of the following gas has a minimum molecular mass?
Oxygen
Nitrogen
Hydrogen
Methane
The total strain energy stored in a body is termed a
Resilience
Proof resilience
Modulus of resilience
Toughness
When the gas is heated at constant pressure, the heat supplied
Increases the internal energy of the gas
Increases the temperature of the gas
Does some external work during expansion
Both (B) and (C)
The layer at the centre of gravity of the beam as shown in the below figure, will be

In tension
In compression
Neither in tension nor in compression
None of these
The mass of flue gas per kg of fuel is the ratio of the
Mass of oxygen in 1 kg of flue gas to the mass of oxygen in 1 kg of fuel
Mass of oxygen in 1 kg of fuel to the mass of oxygen in 1 kg of flue gas
Mass of carbon in 1 kg of flue gas to the mass of carbon in 1 kg of fuel
Mass of carbon in 1 kg of fuel to the mass of carbon in 1 kg of flue gas
For a beam, as shown in the below figure, when the load W is applied in the centre of the beam, the maximum deflection is
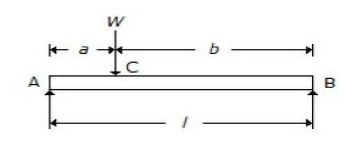
Wl3 / 48EI
5Wl3 / 384EI
Wl3 / 392EI
Wl3 / 384EI
In the tensile test, the phenomenon of slow extension of the material, i. e. stress increasing with the time at a constant load is called
Creeping
Yielding
Breaking
Plasticity
A boiler shell 200 cm diameter and plate thickness 1.5 cm is subjected to internal pressure of 1.5 MN/m, and then the hoop stress will be
30 MN/m²
50 MN/m²
100 MN/m²
200 MN/m²
In a prismatic member made of two materials so joined that they deform equally under axial stress, the unit stresses in two materials are
Equal
Proportional to their respective moduli of elasticity
Inversely proportional to their moduli of elasticity
Average of the sum of moduli of elasticity
The property of a material which allows it to be drawn into a smaller section is called
Plasticity
Ductility
Elasticity
Malleability
The heat and work are mutually convertible. This statement is called __________ law of thermodynamics.
Zeroth
First
Second
Third
Strain is equal to (where l = Original length, and δl = Change in length)
l/δl
δl/l
l.δl
l + δl
In an extensive property of a thermodynamic system
Extensive heat is transferred
Extensive work is done
Extensive energy is utilised
None of these
When a gas is heated at constant pressure
Its temperature will increase
Its volume will increase
Both temperature and volume will increase
Neither temperature not volume will increase
When a body is subjected to a direct tensile stress (σ) in one plane, then maximum normal stress occurs at a section inclined at __________ to the normal of the section.
0°
30°
45°
90°
In compression test, the fracture in cast iron specimen would occur along
The axis of load
An oblique plane
At right angles to the axis of specimen
Would not occur
Brayton cycle consists' of following four processes
Two isothermals and two isentropic
Two isentropic and two constant volumes
Two isentropic, one constant volume and one constant pressure
Two isentropic and two constant pressures
A simply supported beam with a gradually varying load from zero at B and w per unit length at A is shown in the below figure. The shear force at B is equal to
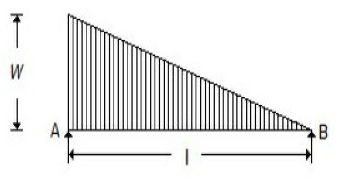
wl/6
wl/3
wl
2wl/3
The distillation carried out in such a way that the liquid with the lowest boiling point is first evaporated and recondensed, then the liquid with the next higher boiling point is then evaporated and recondensed, and so on until all the available liquid fuels are separately recovered in the sequence of their boiling points. Such a process is called
Cracking
Carbonisation
Fractional distillation
Full distillation
The heat and mechanical energies are mutually convertible. This statement was established by
Boyle
Charles
Joule
None of these
Which is the incorrect statement about Carnot cycle?
It is used as the alternate standard of comparison of all heat engines.
All the heat engines are based on Carnot cycle.
It provides concept of maximising work output between the two temperature limits.
All of the above
The bending moment of a cantilever beam of length l and carrying a uniformly distributed load of w per unit length is __________ at the fixed end.
wl/4
wl/2
wl
wl²/2
In an isothermal process,
There is no change in temperature
There is no change in enthalpy
There is no change in internal energy
All of these
The expansion ratio (r) is the ratio of (where v1 = Volume at the beginning of expansion, and v2 = Volume at the end of expansion)
v1/v2
v2/v1
(v1 + v2)/v1
(v1 + v2)/v2
Which of the following is an irreversible cycle?
Carnot
Stirling
Ericsson
None of the above
Youngs modulus of a wire is defined as the stress which will increase the length of wire compared to its original length by
Half
Same amount
Double
One-fourth
The absolute zero temperature is taken as
237°C
-273°C
-237°C
273°C
