Brayton cycle consists' of following four processes
Two isothermals and two isentropic
Two isentropic and two constant volumes
Two isentropic, one constant volume and one constant pressure
Two isentropic and two constant pressures
Correct Answer :
D. Two isentropic and two constant pressures
Related Questions
The value of one bar (in S. I. units) is equal to
1 × 102 N/m2
1 × 103 N/m2
1 × 104 N/m2
1 × 105 N/m2
The radius of the Mohrs circle in the given figure is equal to
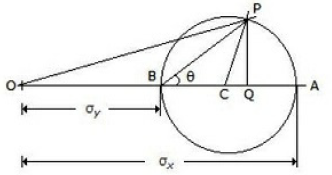
Sum of two principal stresses
Difference of two principal stresses
Half the sum of two principal stresses
Half the difference of two principal stresses
When a bar is cooled to - 5°C, it will develop
No stress
Shear stress
Tensile stress
Compressive stress
The ratio of direct stress to volumetric strain in case of a body subjected to three mutually perpendicular stresses of equal intensity, is equal to
Young's modulus
Bulk modulus
Modulus of rigidity
Modulus of elasticity
In case of an under-reinforced beam, the depth of actual neutral axis is __________ that of the critical neutral axis.
Same as
Less than
Greater than
None of these
The polytropic index (n) is given by
log (p1p2)/log (v1v2)
log (p2/ p1)/log (v1/ v2)
log (v1/ v2)/ log (p1/p2)
log [(p1v1)/(p2v2)]
According to Kelvin-Planck's statement, a perpetual motion of the __________ is impossible.
First kind
Second kind
Third kind
None of these
The atomic mass of nitrogen is __________ oxygen.
Equal to
Less than
More than
None of these
The neutral axis of a transverse section of a beam passes through the centre of gravity of the section and is
In the vertical plane
In the horizontal plane
In the same plane in which the beam bends
At right angle to the plane in which the beam bends
Energy can neither be created nor destroyed, but it can be transformed from one form to another. This statement is known as
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
Kinetic theory of gases
The absolute zero pressure will be
When molecular momentum of the system becomes zero
At sea level
At the temperature of - 273 K
At the centre of the earth
A body is subjected to a tensile stress of 1200 MPa on one plane and another tensile stress of 600 MPa on a plane at right angles to the former. It is also subjected to a shear stress of 400 MPa on the same planes. The maximum normal stress will be
400 MPa
500 MPa
900 MPa
1400 MPa
When it is indicated that a member is elastic, it means that when force is applied, it will
Not deform
Be safest
Stretch
Not stretch
The air standard efficiency of an Otto cycle is given by (where r = Compression ratio, and γ = Ratio of specific heats)
1 - rγ - 1
1 + rγ - 1
1 - (1/ rγ - 1)
1 + (1/ rγ - 1)
Which of the following is a proper sequence?
Proportional limit, elastic limit, yielding, failure
Elastic limit, proportional limit, yielding, failure
Yielding, proportional limit, elastic limit, failure
None of the above
The stress necessary to initiate yielding is
Considerably greater than that necessary to continue it
Considerably lesser than that necessary to continue it
Greater than that necessary to stop it
Lesser than that necessary to stop it
The variables which control the physical properties of a perfect gas are
Pressure exerted by the gas
Volume occupied by the gas
Temperature of the gas
All of these
A double strap butt joint with equal straps is
Always in single shear
Always in double shear
Either in single shear or double shear
None of these
If the depth is kept constant for a beam of uniform strength, then its width will vary in proportional to
Bending moment (i.e. M)
Bending moment² (i.e. M²)
Bending moment³ (i.e. M³)
Bending moment⁴ (i.e. M⁴)
When a closely-coiled helical spring of mean diameter (D) is subjected to an axial load (W), the deflection of the spring (δ) is given by (where d = Diameter of spring wire, n = No. of turns of the spring, and C = Modulus of rigidity for the spring material)
WD3n/Cd⁴
2WD3n/Cd⁴
4WD3n/Cd⁴
8WD3n/Cd⁴
The measurement of a thermodynamic property known as temperature is based on
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
None of these
The layer at the centre of gravity of the beam as shown in the below figure, will be

In tension
In compression
Neither in tension nor in compression
None of these
A hollow shaft of same cross-section area as compared to a solid shaft transmit
Same torque
Less torque
More torque
Unpredictable
When shear force at a point is zero, then bending moment is _________ at that point.
Zero
Minimum
Maximum
Infinity
Which of the following statement is wrong?
The closed cycle gas turbine plants are external combustion plants.
In the closed cycle gas turbine, the pressure range depends upon the atmospheric pressure.
The advantage of efficient internal combustion is eliminated as the closed cycle has an external surface.
In open cycle gas turbine, atmosphere acts as a sink and no coolant is required.
Percentage reduction in area performing tensile test on cast iron may be of the order of
50 %
25 %
20 %
30 %
For a simply supported beam of length l, when a concentrated load W is applied in the centre of the beam, the maximum deflection is
5WL³/ 384EI
WL³/384EI
WL³/ 348EI
WL³/ 48EI
For the same compression ratio, the efficiency of dual combustion cycle is
Greater than Diesel cycle and less than Otto cycle
Less than Diesel cycle and greater than Otto cycle
Greater than Diesel cycle
Less than Diesel cycle
Relation between cp and cv is given by (where cp = Specific heat at constant pressure, cv = Specific heat at constant volume, γ = cp/cv, known as adiabatic index, and R = Gas constant)
cv/ cp =R
cp - cv = R
cv = R/ γ-1
Both (B) and (C)
Which is the incorrect statement about Carnot cycle?
It is used as the alternate standard of comparison of all heat engines.
All the heat engines are based on Carnot cycle.
It provides concept of maximising work output between the two temperature limits.
All of the above
