An oxidation process is accompanied with decrease in the
Number of electrons
Oxidation number
Number of ions
All (A), (B) & (C)
Correct Answer :
A. Number of electrons
Related Questions
Baffles provided on the shell side of a shell and tube heat exchanger are meant for
Providing support for the tubes
Improving heat transfer
Both 'a' & 'b'
Preventing the fouling of tubes & stagnation of shell side fluid
A material subjected __________ must have high resilience.
Shock loading
Vibration
Fatigue
Tension
The main charge in blast furnace is usually
Iron ore & coke
Iron ore, coke & air
Limestone, coke & iron ore
Limestone, sand & iron ore
Thermal diffusivity of a substance is proportional to (where, k = Thermal conductivity)
1/k
k
k2
1/k2
The following type of bonding is strongly directional in solids.
Vander Waal's
Ionic
Metallic
Covalent
The units of the rate constant for a second order reaction are
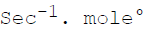
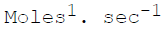
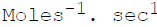
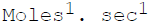
Propulsion of rocket follows from the
Newton's second law of motion
Newton's third law of motion
Law of projectiles
Archimedes principle
In Newton's law of viscosity, which states that the shear stress is proportional to the __________ Co-efficient of viscosity is called dynamic or absolute viscosity(where, V = velocity, 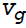 = velocity gradient).
= velocity gradient).
Pick out the wrong statement.
Lead can creep under its own weight at room temperature
The electrical conductivity of gold is considerably reduced by alloying additions due to the decrease in electron movement
Recrystallisation temperature decreases with decrease in strain in a cold worked metal
With increase in temperature, the electrical conductivity of intrinsic semi-conductor will increase
Leakage in a cooking gas cylinder is detected by
Radio-isotopes
Pouring soap solution on the surface and locating the gas bubbles
Halide torch
Pungent smell of mercaptans present in the gas
__________ is the most suitable lubricant for drawing mild steel wires?
Kerosene
Water
Sodium stearate
Lime water
Which of the following is an ore dressing operation?
Classification
Smelting
Roasting
None of these
Production of a hollow product by inflation of a tube or parison is called the __________ process.
Blow moulding
Calendaring
Extrusion
Injection
Pick out the wrong statement.
The X-rays cannot be deflected by electric field unlike cathode rays
The intensity of X-rays can be measured by ionisation current produced due to the ionisation of gas by X-rays
The quality of X-rays can be controlled by varying the anode-cathode voltage
Crystal structure of a material can be studied by an electron microscope
The property of material, by which a given amount of energy is absorbed by it without plastic deformation, is called the
Resilience
Toughness
Ductility
Impact strength
Hydrogen can be
Used in I.C. engines as fuel
Liquefied & stored under cryogenic conditions
Produced by electrolysis of water
All (A), (B) & (C)
The bank of tubes located at the back of the domestic refrigerators are the __________ tubes.
Refrigerant cooling
Evaporator
Condenser
Throttling
A pycnometer is used for the measurement of
Flow rate of molten metal
Specific gravity of a liquid
Composition of alloys
Concentration of a solution
To improve the machinability of steel, it is generally subjected to
Spheroidising
Tempering
Normalising
Annealing
Which of the following has the highest value of refractive index?
Brine (sea water)
Diamond
Distilled water
Glass
In __________ process, there is an increase in entropy with no degradation of energy.
Isothermal expansion
Isochoric heat addition
Polytropic expansion
Isobaric heat addition
Which of the following materials has the poorest electrical conductivity?
Carbon
Aluminium
Silver
Stainless steel
Which of the following is not a principal alloying element for the structural steel?
Molybdenum
Nickel
Manganese
Chromium
For a first order chemical reaction, the concentration of the reactant decreases __________ with time.
Linearly
Exponentially
Logarithmically
Inversely
The heat released by cooling one mole of copper from 400 K to room temperature (300 K) is (assume : Cp of copper is 23 J K-1mole-1)
2300 J
4600 J
230 J
2.3 × 106 J
Atomic __________ of an element is a whole number.
Weight
Number
Volume
Radius
The elastic strain energy of a unit length of an edge dislocation as compared to that of a screw dislocation is
More
Equal
Less
Double
Cassiterite is an ore of
Tin
Lead
Molybdenum
Chromium
'GASOHOL' widely used in Brazil as a motor fuel is a mixture of alcohol and
Petrol
LPG
Light hydrocarbon gases (e.g. butane or propane)
None of these
Which of the following hardness tests does not measure the indentation hardness of metals and alloys?
Vickers hardness test
Shore scleroscope test
Brinell hardness test
Rockwell hardness test
