The point of contra flexure is a point where
Shear force changes sign
Shear force is maximum
Bending moment changes sign
Bending moment is maximum
Correct Answer :
C. Bending moment changes sign
Related Questions
A riveted joint in which every rivet of a row is opposite to other rivet of the outer row, is known as
Chain riveted joint
Diamond riveted joint
Crisscross riveted joint
Zigzag riveted joint
The point of contra flexure is a point where
Shear force changes sign
Shear force is maximum
Bending moment changes sign
Bending moment is maximum
Carnot cycle consists of
Two constant volume and two isentropic processes
Two isothermal and two isentropic processes
Two constant pressure and two isentropic processes
One constant volume, one constant pressure and two isentropic processes
The stress induced in a body due to suddenly applied load compared to when it is applied gradually is
Same
Half
Two times
Four times
Otto cycle efficiency is higher than Diesel cycle efficiency for the same compression ratio and heat input because in Otto cycle
Combustion is at constant volume
Expansion and compression are isentropic
Maximum temperature is higher
Heat rejection is lower
The hard coke is obtained when carbonisation of coal is carried out at
300° to 500°C
500° to 700°C
700° to 900°C
900° to 1100°C
A close cycle gas turbine gives __________ efficiency as compared to an open cycle gas turbine.
Same
Lower
Higher
None of these
The area under the temperature-entropy curve (T - s curve) of any thermodynamic process represents
Heat absorbed
Heat rejected
Either (A) or (B)
None of these
In a reversible adiabatic process, the ratio of T1/T2 is equal to
(p2/p1)γ - 1/ γ
(p1/p2)γ - 1/ γ
(v2/v1)γ - 1/ γ
(v1/v2)γ - 1/ γ
The safe twisting moment for a compound shaft is equal to the
Maximum calculated value
Minimum calculated value
Mean value
Extreme value
The isothermal and adiabatic processes are regarded as
Reversible process
Irreversible process
Reversible or irreversible process
None of these
The ratio of lateral strain to the linear strain within elastic limit is known as
Young's modulus
Bulk modulus
Modulus of rigidity
Poisson's ratio
A shaft revolving at ω rad/s transmits torque (T) in Nm. The power developed is
T.ω watts
2π. T.ω watts
2π. T.ω/75 watts
2π. T.ω/4500 watts
Which of the following statement is wrong?
The closed cycle gas turbine plants are external combustion plants.
In the closed cycle gas turbine, the pressure range depends upon the atmospheric pressure.
The advantage of efficient internal combustion is eliminated as the closed cycle has an external surface.
In open cycle gas turbine, atmosphere acts as a sink and no coolant is required.
Those substances which have so far not been resolved by any means into other substances of simpler form are called
Elements
Compounds
Atoms
Molecules
The natural petroleum may be separated into
Petrol
Kerosene
Fuel oil
Lubricating oil
A series of operations, which takes place in a certain order and restore the initial conditions at the end, is known as
Reversible cycle
Irreversible cycle
Thermodynamic cycle
None of these
Principal planes are planes having
Maximum shear stress
No shear stress
Minimum shear stress
None of the above
One molecule of oxygen is __________ times heavier than the hydrogen atom.
12
14
16
32
When a body is subjected to a direct tensile stress (σ) in one plane, then maximum normal stress occurs at a section inclined at __________ to the normal of the section.
0°
30°
45°
90°
For a beam, as shown in the below figure, the deflection at C is (where E = Young's modulus for the beam material, and I = Moment of inertia of the beam section.
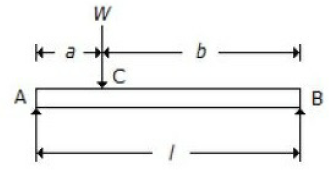
Wl3/48 EI
Wa²b²/3EIl
[Wa/(a√3) x EIl] x (l² - a²)3/2
5Wl3/384 EI
A cycle consisting of __________ and two isothermal processes is known as Stirling cycle.
Two constant pressure
Two constant volume
Two isentropic
One constant pressure, one constant volume
The heat and work are mutually convertible. This statement is called __________ law of thermodynamics.
Zeroth
First
Second
Third
When two bodies are in thermal equilibrium with a third body, they are also in thermal equilibrium with each other. This statement is called
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
Kelvin Planck's law
Which of the following is the correct statement?
For a given compression ratio, both Otto and Diesel cycles have the same efficiency.
For a given compression ratio, Otto cycle is more efficient than Diesel cycle.
For a given compression ratio, Diesel cycle is more efficient than Otto cycle.
The efficiency of Otto or Diesel cycle has nothing to do with compression ratio.
Which of the following cycles has maximum efficiency?
Rankine
Stirling
Carnot
Brayton
One kg of carbon monoxide requires _______kg of oxygen to produce 11/7 kg of carbon dioxide gas.
11/7
9/7
4/7
All of the above
In closed cycle gas turbine, the air is compressed
Isothermally
Isentropically
Polytropically
None of these
A simply supported beam with a gradually varying load from zero at B and w per unit length at A is shown in the below figure. The shear force at B is equal to
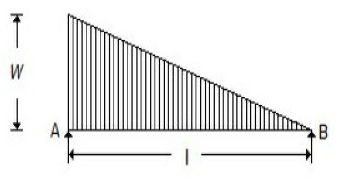
wl/6
wl/3
wl
2wl/3
The thermodynamic difference between a Rankine cycle working with saturated steam and the Carnot cycle is that
Carnot cycle can't work with saturated steam
Heat is supplied to water at temperature below the maximum temperature of the cycle
A Rankine cycle receives heat at two places
Rankine cycle is hypothetical
