The work ratio of a gas turbine plant is given by
(Net work output)/(Workdone by the turbine)
(Net work output)/(Heat supplied)
(Actual temperature drop)/(Isentropic temperature drop)
(Isentropic increase in temperature)/(Actual increase in temperature)
Correct Answer :
A. (Net work output)/(Workdone by the turbine)
Related Questions
The distillation carried out in such a way that the liquid with the lowest boiling point is first evaporated and recondensed, then the liquid with the next higher boiling point is then evaporated and recondensed, and so on until all the available liquid fuels are separately recovered in the sequence of their boiling points. Such a process is called
Cracking
Carbonisation
Fractional distillation
Full distillation
Slenderness of a column is zero when
Ends are firmly fixed
Column is supported on all sides throughout the length
Length is equal to radius of gyration
Length is twice the radius of gyration
According to First law of thermodynamics,
Total internal energy of a system during a process remains constant
Total energy of a system remains constant
Workdone by a system is equal to the heat transferred by the system
Internal energy, enthalpy and entropy during a process remain constant
Which of the following cycles has maximum efficiency?
Rankine
Stirling
Carnot
Brayton
In compression test, the fracture in cast iron specimen would occur along
The axis of load
An oblique plane
At right angles to the axis of specimen
Would not occur
When the gas is heated at constant pressure, the heat supplied
Increases the internal energy of the gas
Increases the temperature of the gas
Does some external work during expansion
Both (B) and (C)
In a free expansion process,
W1 - 2 = 0
Q1 - 2 = 0
dU = 0
All of these
The total strain energy stored in a body is termed a
Resilience
Proof resilience
Modulus of resilience
Toughness
The mass of carbon per kg of flue gas is given by
(11/3) CO2 + (3/7) CO
(3/7) CO2 + (11/3) CO
(7/3) CO2 + (3/11) CO
(3/11) CO2 + (7/3) CO
The maximum bending moment for the beam shown in the below figure, is
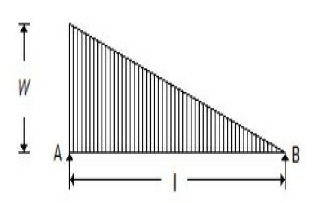
wl²/3√3
wl²/6√3
wl²/9√3
wl²/12√3
The ratio of maximum shear stress developed in a rectangular beam and a circular beam of the same cross-sectional area is
2/3
3/4
1
9/8
For the same compression ratio, the efficiency of Diesel cycle is __________ Otto cycle.
Greater than
Less than
Equal to
None of these
When both ends of a column are fixed, the effective length is
Its own length
Twice its length
Half its length
1/√2 × its length
A cantilever beam is one which is
Fixed at both ends
Fixed at one end and free at the other end
Supported at its ends
Supported on more than two supports
The value of gas constant (R) in S. I. units is
0.287 J/kgK
2.87 J/kgK
28.7 J/kgK
287 J/kgK
If the rivets in adjacent rows are staggered and the outermost row has only one rivets, the arrangement of the rivets is called
Chain riveting
Zigzag riveting
Diamond riveting
Crisscross riveting
The fuel mostly used in cement industry and in metallurgical processes is
Wood charcoal
Bituminous coke
Pulverised coal
Coke
In an isothermal process,
There is no change in temperature
There is no change in enthalpy
There is no change in internal energy
All of these
The point of contraflexure is a point where
Shear force changes sign
Bending moment changes sign
Shear force is maximum
Bending moment is maximum
When two plates are butt together and riveted with cover plates with two rows of rivets, the joint is known as
Lap joint
Butt joint
Single riveted single cover butt joint
Double riveted double cover butt joint
The efficiency of Diesel cycle increases with
Decrease in cut-off
Increase in cut-off
Constant cut-off
None of these
The maximum tangential stress in a thick cylindrical shell is always _________ the internal pressure acting on the shell.
Equal to
Less than
Greater than
None of these
The absolute zero temperature is taken as
237°C
-273°C
-237°C
273°C
A column that fails due to direct stress, is called
Short column
Long column
Weak column
Medium column
One kg of carbon monoxide requires 4/7 kg of oxygen and produces
11/3 kg of carbon dioxide gas
7/3 kg of carbon monoxide gas
11/7 kg of carbon dioxide gas
8/3 kg of carbon monoxide gas
In a tensile test on mild steel specimen, the breaking stress as compared to ultimate tensile stress is
More
Less
Same
More/less depending on composition
If the value of n = 0 in the equation pvn = C, then the process is called
Constant volume process
Adiabatic process
Constant pressure process
Isothermal process
A process, in which the gas is heated or expanded in such a way that the product of its pressure and volume remains constant, is called
Isothermal process
Hyperbolic process
Adiabatic process
Polytropic process
The general law of expansion or compression is pvn = C, The process is said to be hyperbolic, if n is equal to
0
1
γ
∝
A series of operations, which takes place in a certain order and restore the initial conditions at the end, is known as
Reversible cycle
Irreversible cycle
Thermodynamic cycle
None of these
