Pick out the wrong statement.
Cp of monatomic gases such as metallic vapor is about 5 kcal/kg.atom
The heat capacity of solid inorganic substance is exactly equal to the heat capacity of the substance in the molten state
There is an increase in entropy, when a spontaneous change occurs in an isolated system
At absolute zero temperature, the heat capacity for many pure crystalline substances is zero
Correct Answer :
B. The heat capacity of solid inorganic substance is exactly equal to the heat capacity of the substance in the molten state
Related Questions
In the reaction, represented by, 2SO2 + O2 2SO3; ΔH = - 42 kcal; the forward reaction will be favoured by
Low temperature
High pressure
Both (A) and (B)
Neither (A) nor (B)
Which of the following units is not present in both the vapor compression refrigeration system and absorption refrigeration system?
Expansion valve
Condenser
Refrigerator
Compressor
Which of the following is not a common refrigerant?
Freon-12
Ethylene
Ammonia
Carbon dioxide
Pick out the correct statement:
In an isothermal system, irreversible work is more than reversible work
Under reversible conditions, the adiabatic work is less than isothermal work
Heat, work, enthalpy and entropy are all 'state functions'
Matter and energy cannot be exchanged with the surroundings in a closed system
For an ideal solution, the value of activity co-efficient is
0
1
< 1
> 1
A cylinder contains 640 gm of liquid oxygen. The volume occupied (in litres) by the oxygen, when it is released and brought to standard conditions (0°C, 760 mm Hg) will be __________ litres.
448
224
22.4
Data insufficient; can't be computed
Joule-Thomson effect i.e., a throttling process is a constant __________ process.
Entropy
Temperature
Internal energy
Enthalpy
Which of the following is a widely used refrigerant in vapour compression refrigeration system (using large centrifugal compressor)?
Freon
Liquid sulphur dioxide
Methyl chloride
Ammonia
Heat evolved/absorbed during conversion of a substance from one allotropic form to another is termed as the heat of
Fusion
Vaporisation
Transition
None of these
Pick out the wrong statement:
The expansion of a gas in vacuum is an irreversible process
An isometric process is a constant pressure process
Entropy change for a reversible adiabatic process is zero
Free energy change for a spontaneous process is negative
With increase in compression ratio, the efficiency of the otto engine
Increases
Decreases
Remain constant
Increases linearly
Trouton's ratio of __________ liquids is calculated using Kistyakowsky equation.
Polar
Non-polar
Both (A) & (B)
Neither (A) nor (B)
(1/V) (∂V/∂T)P is the mathematical expression
Joule-Thomson co-efficient
Specific heat at constant pressure (Cp)
co-efficient of thermal expansion
Specific heat at constant volume (CV)
The quantitative effect of temperature on chemical equilibrium is given by the
Vant-Hoff equation
Le-Chatelier's principle
Arrhenius equation
None of these
Melting of wax is accompanied with __________ in entropy.
Increase
Decrease
No change
None of these
Throttling (Joule-Thomson effect) process is a constant __________ process.
Enthalpy
Entropy
Pressure
None of these
(∂E/∂T)V is the mathematical expression for
CV
Enthalpy change
Free energy change
None of these
What is the number of degree of freedom for a system of two miscible non-reacting species in vapor-liquid equilibrium forming an azeotrope?
0
2
1
3
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
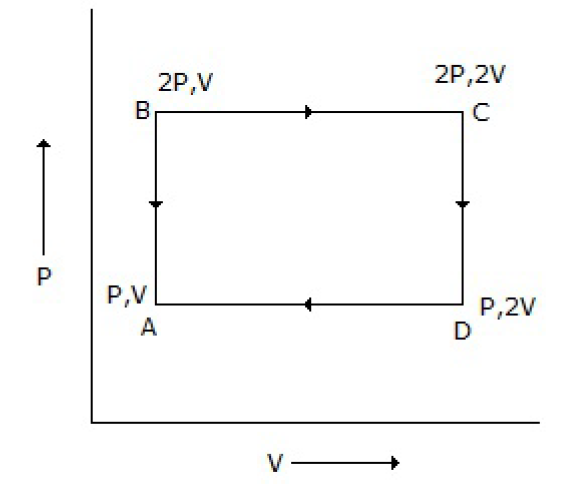
PV
2PV
PV/2
0
The ratio of equilibrium constants (Kp2/Kp1) at two different temperatures is given by
(R/ΔH) (1/T1 - 1/T2)
(ΔH/R) (1/T1 - 1/T2)
(ΔH/R) (1/T2 - 1/T1)
(1/R) (1/T1 - 1/T2)
The heat capacities for the ideal gas state depend upon the
Pressure
Temperature
Both (A) & (B)
Neither (A) nor (B)
Linde gas liquefaction process employs cooling
By throttling
By expansion in an engine
At constant pressure
None of these
Rotary lime kiln is an example of a/an __________ system.
Closed
Open
Isolated
Non-thermodynamic
A solid metallic block weighing 5 kg has an initial temperature of 500°C. 40 kg of water initially at 25°C is contained in a perfectly insulated tank. The metallic block is brought into contact with water. Both of them come to equilibrium. Specific heat of block material is 0.4 kJ.kg-1. K-1. Ignoring the effect of expansion and contraction and also the heat capacity to tank, the total entropy change in kJ.kg-1, K-1 is
-1.87
0
1.26
3.91
Gibbs free energy of a pure fluid approaches __________ as the pressure tends to zero at constant temperature.
Infinity
Minus infinity
Zero
None of these
4 kg moles of an ideal gas expands in vacuum spontaneously. The work done is
4 J
∞
0
8 J
The root mean square speed of molecules of a gas is equal to (where, m = mass of the molecule K = Boltzmanns constant, T = absolute temperature)
√(2KT/m)
√(3KT/m)
√(6KT/m)
3KT/m
Tea kept in a thermos flask is vigorously shaken. If the tea is considered as a system, then its temperature will
Increase
Decrease
Remain unchanged
First fall and then rise
Refrigerants commonly used for domestic refrigerators are
Ethyl chloride or methyl chloride
Freon-12
Propane
NH3 or CO2
In the equation, PVn = Constant, if the value of n = 0, then it represents a reversible __________ process.
Isobaric
Isothermal
Isentropic
Isometric
