The hyperbolic process is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
Correct Answer :
A. Boyle's law
Related Questions
When two bodies are in thermal equilibrium with a third body, they are also in thermal equilibrium with each other. This statement is called
Zeroth law of thermodynamics
First law of thermodynamics
Second law of thermodynamics
Kelvin Planck's law
The columns whose slenderness ratio is less than 80, are known as
Short columns
Long columns
Weak columns
Medium columns
Charles' law states that all perfect gases change in volume by __________ of its original volume at 0°C for every 1°C change in temperature, when pressure remains constant.
1/27th
1/93th
1/173th
1/273th
An open cycle gas turbine works on
Carnot cycle
Otto cycle
Joule's cycle
Stirling cycle
A bar of copper and steel form a composite system, which is heated to a temperature of 40°C. The stress induced in the copper bar will be
Tensile
Compressive
Shear
Zero
The general gas energy equation is (where Q1 - 2 = Heat supplied, dU = Change in internal energy, and W1 - 2 = Work done in heat units)
Q1 - 2 = dU + W1 - 2
Q1 - 2 = dU - W1 - 2
Q1 - 2 = dU/W1 - 2
Q1 - 2 = dU × W1 - 2
Euler's formula holds good only for
Short columns
Long columns
Both short and long columns
Weak columns
High air-fuel ratio in gas turbines
Increases power output
Improves thermal efficiency
Reduces exhaust temperature
Do not damage turbine blades
A series of operations, which takes place in a certain order and restore the initial conditions at the end, is known as
Reversible cycle
Irreversible cycle
Thermodynamic cycle
None of these
A material obeys hooks law up to
Plastic limit
Elastic limit
Yield point
Limit of proportionality
Resilience of a material is considered when it is subjected to
Frequent heat treatment
Fatigue
Creep
Shock loading
Brayton cycle consists' of following four processes
Two isothermals and two isentropic
Two isentropic and two constant volumes
Two isentropic, one constant volume and one constant pressure
Two isentropic and two constant pressures
The efficiency of a gas turbine is given by
(Net work output)/(Workdone by the turbine)
(Net work output)/(Heat supplied)
(Actual temperature drop)/(Isentropic temperature drop)
(Isentropic increase in temperature)/(Actual increase in temperature)
The shear force diagram for a simply supported beam carrying a uniformly distributed load of w per unit length, consists of
One right angled triangle
Two right angled triangles
One equilateral triangle
Two equilateral triangles
An adiabatic process is one in which
No heat enters or leaves the gas
The temperature of the gas changes
The change in internal energy is equal to the mechanical workdone
All of the above
The unit of energy is S. I. units is
Joule (J)
Joule metre (Jm)
Watt (W)
Joule/metre (J/m)
Which of the following cycles has maximum efficiency?
Rankine
Stirling
Carnot
Brayton
The pull required to crush the rivet per pitch length is
p.t.σt
d.t.σc
π/4 × d² × σt
π/4 × d² × σc
In the below figure, the stress corresponding to point D is
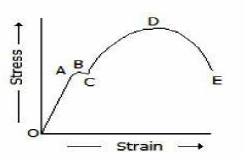
Yield point stress
Breaking stress
Ultimate stress
Elastic limit
Which of the following is a reversible non-flow process?
Isochoric process
Isobaric process
Hyperbolic process
All of these
The total elongation produced in a bar of uniform section hanging vertically downwards due to its own weight is equal to that produced by a weight
Of same magnitude as that of bar and applied at the lower end
Half the weight of bar applied at lower end
Half of the square of weight of bar applied at lower end
One fourth of weight of bar applied at lower end
During a tensile test on a specimen of 1 cm cross-section, maximum load observed was 8 tonnes and area of cross-section at neck was 0.5 cm². Ultimate tensile strength of specimen is
4 tonnes/ cm²
8 tonnes/ cm²
16 tonnes/ cm²
22 tonnes/ cm²
After reaching the yielding stage while testing a mild steel specimen, strain
Becomes constant
Starts decreasing
Increases without any increase in load
None of the above
In a belt drive, the pulley diameter is doubled, the belt tension and pulley width remaining same. The changes required in key will be
Increase key length
Increase key depth
Increase key width
Double all the dimensions
One kg of carbon monoxide requires __________ kg of oxygen to produce 11/7 kg of carbon dioxide gas.
4/7
11/4
9/7
All of these
The gas constant (R) is equal to the __________ of two specific heats.
Sum
Difference
Product
Ratio
According to Avogadro's law, the density of any two gases is __________ their molecular masses, if the gases are at the same temperature and pressure.
Equal to
Directly proportional to
Inversely proportional to
None of these
Proof resilience per material is known as
Resilience
Proof resilience
Modulus of resilience
Toughness
The weakest section of a diamond riveting is the section which passes through
The first row
The second row
The central row
One rivet hole of the end row
The area under the temperature-entropy curve (T - s curve) of any thermodynamic process represents
Heat absorbed
Heat rejected
Either (A) or (B)
None of these
