Which of the following statement is wrong?
The deformation of the bar per unit length in the direction of the force is called linear strain.
The Poisson's ratio is the ratio of lateral strain to the linear strain.
The ratio of change in volume to the original volume is called volumetric strain.
The bulk modulus is the ratio of linear stress to the linear strain.
Correct Answer :
D. The bulk modulus is the ratio of linear stress to the linear strain.
Related Questions
When a bar is subjected to a change of temperature and its deformation is prevented, the stress induced in the bar is
Tensile stress
Compressive stress
Shear stress
Thermal stress
The compression ratio for petrol engines is
3 to 6
5 to 8
10 to 20
15 to 30
The maximum tangential stress in a thick cylindrical shell is always _________ the internal pressure acting on the shell.
Equal to
Less than
Greater than
None of these
The fuel mostly used in steam boilers is
Brown coal
Peat
Coking bituminous coal
Non-coking bituminous coal
When both ends of a column are fixed, the effective length is
Its own length
Twice its length
Half its length
1/√2 × its length
Petrol is distilled at
65° to 220°C
220° to 345°C
345° to 470°C
470° to 550°C
High air-fuel ratio in gas turbines
Increases power output
Improves thermal efficiency
Reduces exhaust temperature
Do not damage turbine blades
An open system is one in which
Heat and work crosses the boundary of the system, but the mass of the working substance does not crosses the boundary of the system
Mass of the working substance crosses the boundary of the system but the heat and work does not crosses the boundary of the system
Both the heat and work as well as mass of the working substance crosses the boundary of the system
Neither the heat and work nor the mass of the working substance crosses the boundary of the system
In a steady flow process, the ratio of
Heat transfer is constant
Work transfer is constant
Mass flow at inlet and outlet is same
All of these
The variables which control the physical properties of a perfect gas are
Pressure exerted by the gas
Volume occupied by the gas
Temperature of the gas
All of these
Which of the following statement is correct?
The increase in entropy is obtained from a given quantity of heat at a low temperature.
The change in entropy may be regarded as a measure of the rate of the availability or unavailability of heat for transformation into work.
The entropy represents the maximum amount of work obtainable per degree drop in temperature.
All of the above
The amount of heat required to raise the temperature of __________ water through one degree is called kilojoules.
1 g
10 g
100 g
1000 g
When a closely-coiled helical spring of mean diameter (D) is subjected to an axial load (W), the stiffness of the spring is given by
Cd⁴/D3n
Cd⁴/2D3n
Cd⁴/4D3n
Cd⁴/8D3n
The stress induced in a body due to suddenly applied load compared to when it is applied gradually is
Same
Half
Two times
Four times
According to Avogadro's law, the density of any two gases is __________ their molecular masses, if the gases are at the same temperature and pressure.
Equal to
Directly proportional to
Inversely proportional to
None of these
One reversible heat engine operates between 1600 K and T2 K and another reversible heat engine operates between T2 K and 400 K. If both the engines have the same heat input and output, then temperature T2 is equal to
800 K
1000 K
1200 K
1400 K
The unit of Young's modulus is
mm/mm
kg/cm
Kg
kg/cm²
Which of the following statement is incorrect?
The liquid fuels consist of hydrocarbons.
The liquid fuels have higher calorific value than solid fuels.
The solid fuels have higher calorific value than liquid fuels.
A good fuel should have low ignition point.
The efficiency of Diesel cycle depends upon
Temperature limits
Pressure ratio
Compression ratio
Cut-off ratio and compression ratio
A process of heating crude oil to a high temperature under a very high pressure to increase the yield of lighter distillates, is known as
Cracking
Carbonisation
Fractional distillation
Full distillation
The heating of gas at constant volume is governed by
Boyle's law
Charles' law
Gay-Lussac law
Avogadro's law
When a body is subjected to a direct tensile stress (σx) in one plane accompanied by a simple shear stress (τxy), the maximum normal stress i
(σx/2) + (1/2) × √(σx² + 4 τ²xy)
(σx/2) - (1/2) × √(σx² + 4 τ²xy)
(σx/2) + (1/2) × √(σx² - 4 τ²xy)
(1/2) × √(σx² + 4 τ²xy)
The ratio of specific heat at constant pressure (cp) and specific heat at constant volume (cv) is always __________ one.
Equal to
Less than
Greater than
None of these
The heat absorbed or rejected by the working substance is given by (where ds = Increase or decrease of entropy, T = Absolute temperature, and dQ = Heat absorbed or rejected)
δQ = T.ds
δQ = T/ds
dQ = ds/T
None of these
The state of stress at a point in a loaded member is shown in the below figure. The magnitude of maximum shear stress is
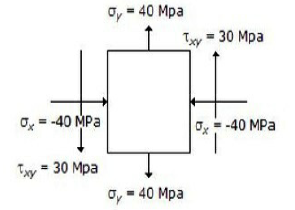
10 MPa
30 MPa
50 MPa
100 MPa
The __________ states that change of internal energy of a perfect gas is directly proportional to the change of temperature.
Boyle's law
Charle's law
Gay-Lussac law
Joule's law
The process is adiabatic, if the value of n in the equation pvn = C, is
0
1
γ
∝
The property of a working substance which increases or decreases as the heat is supplied or removed in a reversible manner, is known as
Enthalpy
Internal energy
Entropy
External energy
A cycle consisting of __________ and two isothermal processes is known as Stirling cycle.
Two constant pressure
Two constant volume
Two isentropic
One constant pressure, one constant volume
If the depth is kept constant for a beam of uniform strength, then its width will vary in proportional to
Bending moment (i.e. M)
Bending moment² (i.e. M²)
Bending moment³ (i.e. M³)
Bending moment⁴ (i.e. M⁴)
