The expression, nRT ln(P1/P2), is for the __________of an ideal gas.
Compressibility
Work done under adiabatic condition
Work done under isothermal condition
Co-efficient of thermal expansion
Correct Answer :
C. Work done under isothermal condition
Related Questions
In case of an __________ process, the temperature of the system increases.
Isothermal compression
Isothermal expansion
Adiabatic expansion
Adiabatic compression
A domestic refrigerator has a/an __________ cooled condenser.
Water
Air
Evaporative
Gas
Heat of formation of an element in its standard state is
0
< 0
> 0
A function of pressure
The expression, nCv(T2 - T1), is for the __________ of an ideal gas.
Work done under adiabatic condition
Co-efficient of thermal expansion
Compressibility
None of these
Solid and liquid phases of a substance are in equilibrium at the
Critical temperature
Melting point
Freezing point
Both (B) and (C)
In an ideal gas mixture, fugacity of a species is equal to its
Vapor pressure
Partial pressure
Chemical potential
None of these
The compressibility factor for an ideal gas is 1. Its value for any other real gas is
1
< 1
> 1
Either (B) or (C), depends on the nature of the gas
In a turbine, the fluid expands almost
Isothermally
Isobarically
Adiabatically
None of these
Isotherm on an enthalpy-concentration diagram, for an ideal solution will be a
Straight line
Sine curve
Parabola
Hyperbola
For a real gas, the chemical potential is given by
RT d ln P
RT d ln f
R d ln f
None of these
What is the number of degrees of freedom for liquid water in equilibrium with a mixture of nitrogen and water vapor?
2
0
3
1
A system is said to be isopiestic, if there is no __________ change.
Temperature
Pressure
Volume
None of these
The temperature at which a real gas obeys the ideal gas laws over a wide range of pressure is called the __________ temperature.
Critical
Boyle
Inversion
Reduced
First law of thermodynamics is mathematically stated as
dQ = dE + dW
dQ = dE - dW
dE = dQ + dW
dW = dQ + dE
A system is said to be at equilibrium, if the entropy of the system has reached __________ value.
Minimum
Zero
Maximum
None of these
Fundamental principle of refrigeration is based on the __________ law of thermodynamics.
Zeroth
First
Second
Third
Pick out the wrong statement.
Minimum number of degree of freedom of a system is zero
Degree of freedom of a system containing a gaseous mixture of helium, carbon dioxide and hydrogen is 4
For a two phase system in equilibrium made up of four non-reacting chemical species, the number of degrees of freedom is 4
Enthalpy and internal energy change is zero during phase change processes like melting, vaporisation and sublimation
Throttling (Joule-Thomson effect) process is a constant __________ process.
Enthalpy
Entropy
Pressure
None of these
Which of the following identities can be most easily used to verify steam table data for superheated steam?
(∂T/∂V)S = (∂p/∂S)V
(∂T/∂P)S = (∂V/∂S)P
(∂P/∂T)V = (∂S/∂V)T
(∂V/∂T)P = -(∂S/∂P)T
The most important application of distribution law is in
Evaporation
Liquid extraction
Drying
Distillation
Extensive properties of a thermodynamic system depend upon the __________ of the system.
Specific volume
Temperature
Mass
Pressure
A refrigeration cycle is the same as a __________ cycle,
Turbine
Heat engine
Reversed heat engine
None of these
The change in __________ is equal to the reversible work for compression in steady state flow process under isothermal condition.
Internal energy
Enthalpy
Gibbs free energy
Helmholtz free energy
Enthalpy changes over a constant pressure path are always zero for __________ gas.
Any
A perfect
An easily liquefiable
A real
Equation which relates pressure, volume and temperature of a gas is called the
Equation of state
Gibbs Duhem equation
Ideal gas equation
None of these
For a single component two phase mixture, the number of independent variable properties are
Two
One
Zero
Three
An ideal monatomic gas is taken round the cycle ABCDA as shown below in the P-V diagram. The work done during the cycle is
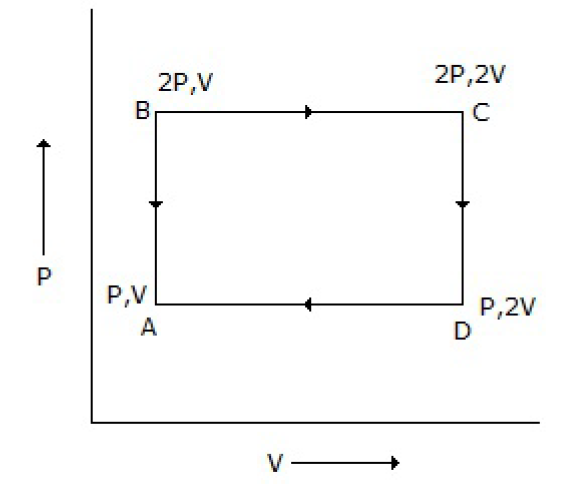
PV
2PV
PV/2
0
PVγ = Constant (where, γ = Cp/Cv) is valid for a/an __________ process.
Isothermal
Isentropic
Isobaric
Adiabatic
Lowering of condenser temperature (keeping the evaporator temperature constant) in case of vapour compression refrigeration system results in
Increased COP
Same COP
Decreased COP
Increased or decreased COP; depending upon the type of refrigerant
Dry ice is
Moisture free ice
Solid helium
Solid carbon dioxide
None of these
