The value of gas constant 'R' is
1.987 cal/gm mole °K
1.987 BTU/lb. mole °R
Both (A) and (B)
Neither (A) nor (B)
Correct Answer :
C. Both (A) and (B)
Related Questions
Internal energy of an element at 1 atm and 25° C is __________ kcal/kg.mole.
0
273
25
None of these
The ratio of equilibrium constants (Kp2/Kp1) at two different temperatures is given by
(R/ΔH) (1/T1 - 1/T2)
(ΔH/R) (1/T1 - 1/T2)
(ΔH/R) (1/T2 - 1/T1)
(1/R) (1/T1 - 1/T2)
For an irreversible process involving only pressure-volume work
(dF)T, p <0
(dF)T, p = 0
(dF)T, p > 0
(dA)T, v >0
For an ideal gas, the internal energy depends upon its __________ only.
Molecular size
Temperature
Volume
Pressure
An isentropic process is carried out at constant
Volume
Pressure
Temperature
All (A), (B) and (C)
Trouton's ratio of __________ liquids is calculated using Kistyakowsky equation.
Polar
Non-polar
Both (A) & (B)
Neither (A) nor (B)
The chemical potential of any constituent of an ideal solution depends on the __________ of the solution.
Temperature
Pressure
Composition
All (A), (B) and (C)
In the reaction, C + O2 → CO2; ΔH = - 94 kcal. What is the heat content (enthalpy) of O2?
-94 kcal
> -94 kcal
< - 94 kcal
Zero
Entropy change of the reaction, H2O (liquid) → H2O (gas), is termed as the enthalpy of
Solution
Vaporisation
Formation
Sublimation
The compressibility factor for an ideal gas is 1. Its value for any other real gas is
1
< 1
> 1
Either (B) or (C), depends on the nature of the gas
Work done in case of free expansion is
Indeterminate
Zero
Negative
None of these
Number of phases in a colloidal system is:
1
2
3
4
For the gaseous phase chemical reaction, C2H4(g) + H2O(g) ↔ C2H5OH(g), the equilibrium conversion does not depend on the
Steam to ethylene ratio
Temperature
Pressure
None of these
Free energy
Decreases in all spontaneous (or irreversible) processes
Change during a spontaneous process has a negative value
Remains unchanged in reversible processes carried at constant temperature and pressure
All (A), (B) and (C)
Sound waves propagation in air exemplifies an __________ process.
Adiabatic
Isothermal
Isometric
None of these
Degree of freedom of the system ice-watervapour will be
0
1
2
3
Third law of thermodynamics is helpful in
Prediction of the extent of a chemical reaction
Calculating absolute entropies of substances at different temperature
Evaluating entropy changes of chemical reaction
Both (B) and (C)
y = specific heat ratio of an ideal gas is equal to
Cp/Cv
Cp/(CP-R)
1 + (R/CV)
All (A), (B) and (C)
Out of the following refrigeration cycles, which one has maximum COP?
Air cycle
Carnot cycle
Ordinary vapor compression cycle
Vapor compression with a reversible expansion engine
The chemical potential of a component (μi) of a phase is the amount by which its capacity for doing all work, barring work of expansion is increased per unit amount of substance added for an infinitesimal addition at constant temperature and pressure. It is given by
(∂E/∂ni)S, v, nj
(∂G/∂ni)T, P, nj = (∂A/∂ni) T, v, nj
(∂H/∂ni)S, P, nj
All (A), (B) and (C)
The number of degrees of freedom for an azeotropic mixture in a two component vapour-liquid equilibria is/are
Zero
One
Two
Three
Equation which relates pressure, volume and temperature of a gas is called the
Equation of state
Gibbs Duhem equation
Ideal gas equation
None of these
Consider the process A & B shown in the figure given below: In this case, it is possible that
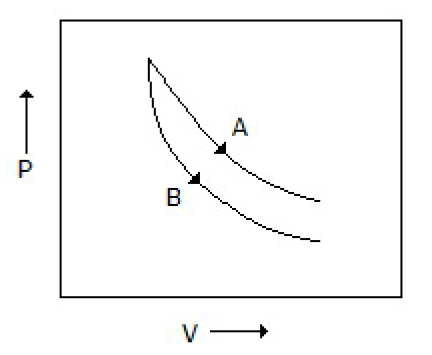
Both the processes are adiabatic
Both the processes are isothermal
Process A is isothermal while B is adiabatic
Process A is adiabatic while B is isothermal
Pick out the wrong statement pertaining to the decomposition of PCl5 represented by, PCl5 PCl3 + Cl2.Degree of dissociation of PCl5 will
Decrease on addition of Cl2
Increase on addition of an inert gas at constant pressure
Decrease on increasing the pressure of the system
None of these
At triple point (for one component system), vapour pressure of solid as compared to that of liquid will be
More
Less
Same
More or less; depending on the system
In which of the following reaction equilibrium, the value of equilibrium constant Kp will be more than is Kc?
2HI H2 + I2
N2O4 2NO2
2SO2 + O2 2SO3
None of these
Entropy change for an irreversible process taking system and surrounding together is
0
> 0
< 0
None of these
Compressibility factor (i.e., the ratio of actual volume of gas to the volume predicted by ideal gas law) for all gases are
Always greater than one
Same at the same reduced temperature
Same at the same reduced pressure
Both (B) & (C)
For an ideal solution, the value of activity co-efficient is
0
1
< 1
> 1
Consider the reaction, C + O2 CO2; ΔH = - 94 kcal. What will be the value of ΔH for the reaction CO2 → C + O2?
-94 kcal
+94 kcal
> 94 kcal
< -94 kcal
